 |
de | fr | en print view![]()
Annual report 2006
PDF versionSummary of the Year’s Activities
Improvements to the Foundation’s website
Extensive information about all the Foundation’s activities can be found on its website at www.forschung3r.ch. The homepage was improved during 2006.
20 projects subsidised
A total amount of Fr. 726,100 was paid out for 13 ongoing projects and 7 that were completed during 2006.
6 new projects
Six new projects were approved for funding during the past year for which a total of Fr. 735,000 was earmarked. These new projects are described in detail in the list of funded projects on the Foundation’s website (www.forschung3r.ch/en/projects/index.html).
Development of an in-vitro system for modelling bioaccumulation of neutral, ionizable, and metabolically active organic pullutants in fish (100/06) B. Escher, Dept. of Environmental Toxicology, EAWAG, Dübendorf. The aim of this project is to develop a method of fulfilling the stipulations of OECD guideline 305 concerning the bioaccumulation of chemicals using an adapted artificial membrane system instead of fish.
Organotypic CNS slice cultures as an in-vitro model for immune mediated tissue damage and repair in multiple sclerosis (101/06) N. Goebels, Dept. of Neurology and Neuroimmunology, University Hospital, Zurich. This project aims to use organotypic slice cultures from mouse brain, in conjunction with electrophysiological measurements, to examine certain aspects of multiple sclerosis in vitro in order to alleviate the need for laboratory animals.
Isolated, autologous blood-perfused heart: replacement of heterotopic heart transplantation (102/06) A. Bogdanova, Institute of Veterinary Physiology, University of Zurich. This project aims to develop a heart-lung machine that will enable isolated rodent hearts to be used to investigate pathological processes following ischaemia without using laboratory animals.
An in vitro model of central nervous system infection and regeneration: neuronal stem cells as targets of brain damage and regenerative therapies in bacterial meningitis (103/06) S. Leib, Institute of Infectious Diseases, University of Berne. The aim of this project is to replace live animals by cultured neuronal stem cells and organ slices from rats for examining the cell processes and regenerative therapies in bacterial meningitis.
Development of in vitro strategies to propagate and characterize hemotrophic mycoplasmas (104/06) R. Hofmann-Lehmann, Clinical Laboratory, Vetsuisse Faculty, University of Zurich. Cell culture methods are to be developed in order to avoid using animals to obtain hemoplasmas to examine contagiousness.
Establishment of an in vitro system for the prediction of the degree of virulence of classical swine fever virus isolates (105/06) N. Ruggli, Institute of Virology and Immunoprophylaxis, Mittelhäusern. Here the aim is to predict the degree of virulence in cell cultures instead of using live animals.
4 projects successfully completed
Induction of a primary T-cell mediated immune response against drugs and drug metabolites in vitro (80/01) W. J. Pichler, Dept. for Rheumatology and clin. Immunology/Allergology, University Hospital, Berne. The results show that the immunogenic (and allergenic) potential of a drug can be determined using the T-cell activation test. This represents an important basis for developing a test that could alleviate the need for laboratory animals.
Development of an in vitro culture model to generate Neospora caninum and Toxoplasma gondii oocysts and sporozoites (85/03) A. Hemphill, Institute of Parasitology, Vetsuisse Faculty and Faculty of Medicine, University of Berne. This project has expanded the necessary basic knowledge in this field and opened the way to culturing oozoites in vitro instead of in dogs.
Information on serum-free cell lines: an interactive database (87/03) C. Strebel, CePower GmbH, Wädenswil. The interactive SEFREC database is now accessible on the internet (www.sefrec.com).
Assessing animal health and welfare and recognising pain and distress (88/03) P. Flecknell, Comparative Biology Centre, University of Newcastle, England. This project resulted in an e-learning module: the AHWLA (Assessing the Health and Welfare of Laboratory Animals) (www.ahwla.org.uk). The Foundation used this to create a module on pain recognition for its internet training course (http://3r-training.tierversuch.ch/en/module_3r/pain.html). This uses video sequences and images to enable researchers to train students to recognise pathological pain in laboratory animals so that they will be able to take measures to alleviate pain and distress in good time.
3R-Info-Bulletins
3R-Info bulletins are published on the Foundation’s website (www.forschung3r.ch/en/publications/index.html).
Improving pain therapy in laboratory mice (issue no. 31, January 2006) The problem of pain recognition is defined and addressed on the basis of project 71/00 (Dr. Margarete Arras, Institute for Laboratory Animal Science, University of Zurich); behaviour and physiological and molecular parameters (stress gene) are considered as recognition factors.
The use of non-invasive methods in animal experimentation for examining diseases of the respiratory tract (issue no. 32, May 2006) This article describes how Dr. N. Beckmann (Novartis Pharma, Basle; project 82/02) succeeded in using asthma research to show how the use of MRI can drastically reduce the number of animals required, as well as attenuating stress and shortening the duration of the experiments.
Predicting the allergic reaction to medication in vitro (issue no. 33, September 2006) This feature shows how Prof. W. Pichler (University Hospital, Berne; project 80/01) was able to demonstrate that even substances that are not considered hapten-carrier complexes can stimulate T-cell division, i.e. the substances act as immunogens solely by reason of their chemical structure.
1. Origin of the Foundation
The Foundation is a cooperative institution set up by the Parliamentary Group for Animal Experimentation Questions (public organ), Interpharma (Novartis Pharma Ltd, F. Hoffmann-La Roche Ltd, Serono Ltd and the associated members Actelion Ltd, Cilag Ltd and Vifor Ltd) and the Foundation for Animal-Free Research (animal protection). It was entered in the commercial register on 18th August, 1987.
The funds provided to subsidise research stem from the Federal Veterinary Office and Interpharma.
2. Purpose of the Foundation
The purpose of the 3R Research Foundation Switzerland is to promote alternative research methods which avoid the use of animals, through grants for research projects. The organisation supports first and foremost projects aimed at developing new methods or refining accepted methods (validation) which offer practical improvements vis-à-vis standard animal experimentation in line with the 3R motto Reduce, Refine, Replace.
A broad range of projects is sponsored on the condition that they are likely to reduce the number of animals used or the stress and/or pain suffered. Projects considered must be based on the Foundation’s three principles and are mainly in the bio-medical multidisciplinary field.
3. Organisation of the Foundation
The Administrative Board
The Administrative Board of the Foundation is made up of nine members, three representing the Parliamentary Group for Animal Experimentation Questions, two representing animal protection, two from Interpharma and two from the Federal Veterinary Office. Current members are:
Dr. Hugo Wick, Basle (Chairman)
Christine Egerszegi-Obrist, member of the National Council, Mellingen (Deputy Chairwoman)
Chantal Galladé, member of the National Council, Winterthur
Dr. Peter Bossard, Horw
Dr. Franz P. Gruber, Zurich
Dr. Peter Heer, Corporate Communications, F. Hoffmann-La Roche Ltd., Basle
Prof. Paul Herrling, Head of Research, Novartis International, Basle
Ursula Moser, B.Sc., Federal Veterinary Office, Berne-Liebefeld
Dr. Hans Wyss, Director of the Federal Veterinary Office, Berne-Liebefeld
The Evaluation Committee
Prof. Peter Maier, Uster (as from 1.1.07) (Chairman)Dr. Franziska Boess, F. Hoffmann-La Roche Ltd, Basle
Prof. Kurt Bürki, Institute of Laboratory Animal Science, University of Zurich
Prof. Clemens A. Dahinden, Institute of Immunology and Allergology, University Hospital, Berne
Prof. Max Gassmann, Institute of Veterinary Physiology, University of Zurich (until 2.3.06)
Prof. Marianne Geiser Kamber, Institute of Anatomy, University of Berne
Dr. Franz P. Gruber, Zurich (until 31.12.06)
Dr. Kurt Lingenhöhl, Novartis Pharma AG, 4002 Basle (as from 21.3.06)
Prof. Thomas Lutz, Institute of Veterinary Physiology, University of Zurich
Ursula Moser, B.Sc., Federal Veterinary Office, Berne-Liebefeld
Susanne Scheiwiller, B.Sc., FFVFF, Zurich (as from 1.1.07)
Dr. Alfred Schweizer, Friedrich Miescher-Institute, Basle (until 31.12.06)
Scientific adviser
Prof. Peter Maier, Uster
Auditors
KPMG AG, Gümligen-Berne
Supervisory body
Federal Department of Home Affairs
Articles and statutes of the Foundation
- Deed of foundation dated 13th February, 1987
- Regulations dated 15th May, 1987
- Guidelines for awarding research grants dated 15th May, 1987
4. Activities during 2006
In its twentieth year of existence the Administrative Board met three times, namely in March, June and December, for a half-day meeting. Apart from the statutory business concerning the end of the business year 2005, the Board addressed the following issues.
Research funds for 2006 were allotted to 12 projects already underway. In addition, 6 new projects were approved, while 17 applications were rejected. The Board also took note of the final assessment by the Evaluation Committee of 4 projects which had been completed in the previous years.
In March 2006 discussions focused on the financial statements for 2005 as well as how to mark the 20th anniversary of the Foundation, and the guidelines for allotting research funds in relation to the submission of applications were defined. During its meeting in June the Administrative Board concentrated mainly on the 20th anniversary and the new 3R brochure. It approved the outline for the 3R brochure and gave the go-ahead for signing a contract with the agency in question. Moreover, the Board decided to organise a scientific meeting in autumn 2007 and to mark the occasion by bringing out a special issue of ALTEX describing the sustainability of funding research projects. This issue is to be based on the 3R-Info-bulletins, which will be updated by the various authors. Summaries of current projects are to be published. Finally, the Foundation’s regulations concerning the status and function of the Evaluation Committee were revised. As an independent organ of the Foundation, chaired by the scientific adviser, from 2007 on the Evaluation Committee will assess applications for research grants and pass its recommendations on to the Administrative Board. At the meeting held in december 2006, as well as approving new and completed projects, the Board discussed issues relating to the financial statements for 2006 and the budget for 2007. The Administrative Board accepted the scientific adviser’s report on the Foundation’s presence at various events and thanked him for his commitment.
With the support of the scientific adviser, the Evaluation Committee held two meetings during the year, where in particular they assessed new applications and evaluated completed projects. The voluntary work of the members of the Evaluation Committee in this connection is much appreciated.
The scientific adviser's tasks included publishing the 3R Info Bulletin (as a brochure and on the Foundation’s website at www.forschung3r.ch), writing brief scientific reports in English which present the projects receiving funding and regularly updating the Foundation’s website. He was also kept busy monitoring the 3R Training Course internet learning programme and integrated the new pain recognition module (http://3r-training.tierversuch.ch/en/module_3r/pain.html). In addition, he spent much time – as always – advising applicants and project managers, obtaining intermediate reports, evaluating project outlines, dealing with enquiries and explaining why projects had been rejected. The scientific adviser was also closely involved in the preparations for the new 3R brochure, which is due to come out mid-2007, as well as the scientific meeting planned for autumn 2007 to mark the Foundation’s 20th anniversary. Preparations are also underway for the special issue of ALTEX, due to be published at the end of August 2007. Finally, the scientific adviser represented the Foundation at several scientific meetings in Switzerland and abroad, namely as a member of the board at the Annual Meeting of the European Consensus Platform for 3R Alternatives to Animal Experimentation (http://ecopa.vub.ac.be) in Brussels. Special mention should also be made of his appointment to the Advisory Board of the AcuteTox Consortium Meetings, which met in Ostend in 2006.
5. Projects subsidised
Overview of the number of applications and approvals
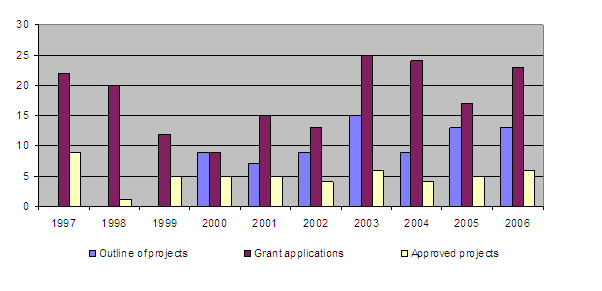
During the year 4 projects were completed (80/01, 85/03, 87/03, 88/03). Together with those projects completed earlier (1-5/87, 6-15/88, 16/89, 17-20/90, 21-24/91, 25-42/92, 43-44/95, 45-55/96, 56-64/97, 65/98, 66-70/99, 71-75/00, 76-79/01, 81/02, 83/02, 86/03) this brings the total of finished projects to 86 out of 105.
3R Training Course
The Foundation has set up the 3R Training Course internet learning programme to offer individual, specialised further training for people who carry out or supervise animal experiments. This course is available in German and English at http://3R-training.tierversuch.ch. Texts, images, links and documents provide visitors to the site with information on alternatives to animal experimentation.
The 3R Training Course has been recognised by the Association of Cantonal Veterinary Surgeons as a further training course within the meaning of the Federal Veterinary Office’s ordinance of 12th October 1998 on the basic and further training of persons involved in animal experimentation (SR 455.171.2). During 2006 fifteen certificates confirming that people had passed the on-line examination were issued.
Quality of the projects
Various factors indicate the quality of the completed and current projects:
Renowned scientists
The specialised knowledge of the project managers and the infrastructure that is available are two key factors. The names of the project managers and institutes involved show that the commitment to the 3R principles has strong support in scientific and research circles. News about academic accolades for our project managers is proof of their ranking. In 2006 Dr. N. Beckmann (project 82/02) became eligible for a professorship and Dr. M. Geiser Kamber and Dr. R. Rieben were awarded honorary professorships by the University of Berne.
Publication of results
The number of publications resulting from a project and the frequency with which they are cited imply that the results obtained are being properly evaluated. A glance at the summaries of the projects shows a satisfactory number of publications that we are expecting or that have already been added to our list.
Awards and public reactions
Various projects funded by the Foundation have already won prizes or been mentioned in the press. In 2006 Prof. D. Hemphill (project 85/03) was awarded the Egon Naef Prize. The publication of T. Kröber and P. M. Guerin’s paper, An in vitro feeding assay to test acaricides for control of hard ticks, in Pest Management Science (63, 17-22, 2006), which resulted from project 79/01, was heralded in the Swiss, German and English press as a breakthrough in replacing animals in research.
6. Personnel
There were some important changes in the Evaluation Committee during 2006. Prof. Max Gassmann, Dr. Franz P. Gruber and Dr. Alfred Schweizer resigned from the Committee and the Foundation expressed its appreciation of their work on its behalf. Dr. Kurt Lingenhöhl (Novartis Pharma AG, Basle) and Susanne Scheiwiller, B.Sc. (co-chair of the Foundation for Animal-Free Research, FFVFF, in Zurich) were elected as new members. Prof. Peter Maier was elected chairman of the Evaluation Committee as from 1 January 2007.
7. 3R-Info-Bulletin
In 2006 three more new 3R-Info bulletins were published with a print-run of 1,000 copies each in English, and distributed among interested parties. The information bulletins are also published on the Foundation’s website (www.forschung3r.ch/en/publications/index.html).
The latest 3R-Info bulletins are:
| No. | Date | Topic |
|---|---|---|
| 34 | January 2007 | Exploring natural anticoagulation by endothelial cells: A novel in vitro model |
| 33 | September 2006 | Predicting drug hypersensitivity by in vitro tests |
| 32 | May 2006 | Non-Invasive Methods: Investigation of Airways Diseases by MRI in Rats |
| 31 | January 2006 | Improvement of Pain Therapy in Laboratory Mice |
List of the other 3R-INFO BULLETINS
8. List of new projects approved in 2006
A complete list of projects with summaries of each can be found on the Foundation’s website (www.forschung3r.ch/en/projects/index.html).
The brief scientific project reports in English, which are updated once a year, also appear on the website and indicate that almost all projects have progressed well. The project managers’ reports tend increasingly to include helpful images. These reports published on the internet are much appreciated by those involved in the research projects as a platform for presenting their work. From the opposite point of view, this system also enables other researchers all over the world to discover new 3R methods without delay.
9. Financial business
A total of some Fr. 753,000 was paid out for research in 2006 (Fr. 726,000 grants to research projects, Fr. 25,000 for the 3R brochure and Fr. 2,000 for participation in conferences). Some further Fr. 110,000 was spent on project monitoring and information, of which Fr. 19,000 served to improve the 3R internet training course. A sum of Fr. 93,000 was spent on administration. Total expenditure therefore amounted to around Fr. 956,000.
Expenditure on current projects (Fr. 726,100) was some Fr. 63,700 over budget (Fr. 662,400); this was principally due to the fact that Fr. 120,000 was paid out for three new projects and the 5% reserve (budgeted at Fr. 66,200) was paid out on numerous projects that were completed, amounting to Fr. 47,400 in all. On the other hand, Fr. 54,000 earmarked for one project was not used because the payments were put in abeyance until certain problems are solved. Only one grant of Fr. 2,000 was applied for in relation to participation in a conference. The total of approximately Fr. 203,400 for project monitoring, information and administration was around Fr. 7,000 over budget (Fr. 196,500), Fr. 11,500 less than budget being spent on the internet training course and Fr. 23,600 more than budget being required for administrative expenses. This excess expenditure was mainly accounted for by costs incurred by the secretariat relating to the additional meeting of the Administrative Board and updating the Foundation’s website. Another item involved preparatory work on the 3R brochure.
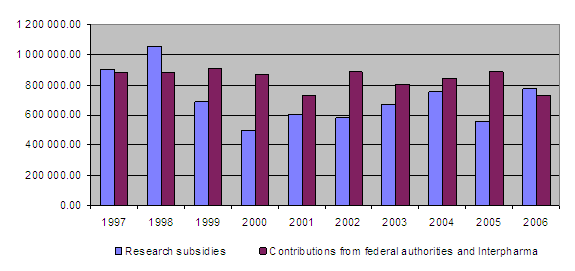
On the income side, the equal financial commitment of the federal authorities and Interpharma represents the basic funding for the Foundation’s activities. The Confederation and Interpharma each provided the Foundation with Fr. 365,000 in 2006. At the end of the year the Federal Veterinary Office promised to donate an additional amount of Fr. 100,000, which was entered at the beginning of 2007. The Foundation will also be applying for a corresponding increase in the amount received from Interpharma in 2007.
Thanks to the rise in interest rates, it was to the Foundation’s advantage to invest cash not required immediately in several different time deposits, which resulted in a considerable rise in interest earned in comparison with the previous year.
Total income was therefore around Fr. 736,100 (funding from the Confederation and Interpharma together being Fr. 730,000, interest earned amounting to Fr. 4,700 and 3R training course fees yielding Fr. 1,400) while total expenditure amounted to Fr. 956,000. This gives an excess of expenditure over income of around Fr. 220,100. The unused contributions item therefore fell from approximately Fr. 757,200 at the end of 2005 to Fr. 537,100 at the end of 2006.
The budget for 2007 includes around Fr. 678,000 for current projects and a maximum amount of Fr. 500,000 for new projects.
Overview of grants awarded between 1987 and 2006
At the end of 2006 a total of Fr. 14,646,735.80 had been granted for projects and other subsidies, of which Fr. 13,397,816,40 has been paid out so far. Together the federal authorities and Interpharma have contributed Fr. 15,888,000 to the Foundation since 1987.
10-year overview
10. Financial statements
| Profit and loss account 2006 | Expenditure | Income | |
|---|---|---|---|
| Income | |||
| Federal contribution | 365 000.00 | ||
| Contribution from Interpharma | 365 000.00 | ||
| Total contributions | 730 000.00 | ||
| Interest on bank account | 4 682.60 | ||
| Research funds repaid | 0.00 | ||
| Other income | 1 471.00 | ||
| Total income | 899 205.73 | ||
Expenditure | |||
| Research grants | 753 139.95 | ||
| Project supervision and information | 109 428.85 | ||
| Administrative expenses | 93 693.50 | ||
| Total expenditure | 956 262.30 | ||
| Excess expenditure over income | - 220 108.70 | ||
| 736 153.60 | |||
| Balance as per 31st December 2006 | Assets | Liabilities | |
| Liquid Assets | |||
| Bank | 577 086.39 | ||
| Accounts payable | 1 638.90 | ||
| Accounting apportionment assets | 27.00 | ||
Liabilities | |||
| Accounting apportionment liabilities | 40 614.20 | ||
| Unused project funds | |||
| Carried forward 1. 1. 2006 | 757 246.79 | ||
| expend. over income | - 220 108.70 | ||
| 537 138.09 | |||
| Capital of the Foundation | 1 000.00 | ||
| 578 752.29 | 578 752.29 | ||
Contingent liabilities | |||
| Approved research grants not yet paid out | Sfr. 1 248 919.40. | ||
Münsingen, 26th March 2007
3R RESEARCH FOUNDATION
President: signed Dr. Hugo Wick
Secretary: signed E. Diener
11. Auditors' report to the Administrative Board
As 3R Research Foundation’s auditors, KPMG AG in Gümligen-Berne has examined the books and the annual financial statements on the basis of current financial reporting standards and recommends that they be approved.
| Last modified 2014/06/04 |