 |
de | fr | en Druckansicht ![]()
3R-INFO-BULLETIN 13
January 2000
The Authors
  |
Dr. Arie Bruinink1 and Prof. Dr. Peter Maier2 were both head of a research group in Cellular Toxicology at the former Institute of Toxicology, ETH Zürich and University of Zürich, Switzerland.
current address:
1 Biocompatible Materials Science and Engineering ETH Zürich, Wagistrasse 23,
CH-8952 Schlieren,
Switzerland
bruinink@biocomp.mat.ethz.ch
2Institute of Toxicology ETH Zürich,
Schorenstr. 16,
CH-8603 Schwerzenbach,
Switzerland
maier@toxi.biol.ethz.ch
Editor
Peter Maier, Scientific Adviser of the 3R Research Foundation
Identification of neurotoxic chemicals in cell cultures
Assessment of neurotoxicity
The identification of potential neurotoxic activity (damaging nervous tissue cells) of chemicals and drugs is an important task within the framework of the toxicity assessment of chemicals. Current guidelines recom-mend the use of animal tests, because of the possibility that a compound might be converted by xenobiotic metabolism to metabolites with neurotoxic activity. Current risk assessment guidelines for neurotoxic organophosphorus compounds (OP) are based on the in vivo hen model due to its high sensitivity.
Conversion of nontoxic chemicals to neurotoxic metabolites.
A chemical might exert its toxicity directly or indirectly, following metabolism in the liver. Stable metabolites can be released from liver cells into the blood stream, transported to the brain and there cross the blood brain-barrier. This pathway has been well established for some OPs, but cannot be reproduced in human or animal brain tumor (neuroblastoma) cell cultures. Accordingly such cell culture systems fail to identify indirectly acting neurotoxic compounds.
 |
| Fig. 1a. Rat hepatocytes (4 days in culture) |
 |
| Fig. 1b. Chicken brain cells (8 days in culture). |
The 3R goals of the project
The goal of the project was to assess the possibility of identifying chemicals with a Iiver-mediated brain toxicity in a cell culture system[*]. Cocultures were established between primary rat hepatocytes (1) and chicken brain cells isolated from total brain of Tetra SL embryonic chicken at stage 29 (2). The hepatocytes (Fig 1a) represent the liver, the embryonic brain cells (Fig.1b) the ultimate target. Stable hepatic metabolites in the supernatant have a high chance of reaching brain tissue via the blood circulation. The test compound can be added either directly to the cocultures (Fig.2), or first to hepatocyte cultures, following which the supernatant is transferred to the brain cells (Fig.2). Comparison between the two results provides information on the stability of the hepatocyte-derived metabolites.
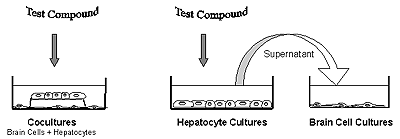 |
| Fig. 2. Exposure of brain cells to liver cell mediated metabolites in cocultures or via supernatant. |
Brain cell toxicity was assessed by measuring the bioreductive capacity (conversion of the dye MTT), the lysososmal activity (neutral red uptake) and the brain specific acetycholinesterase (AchE) activity of the cells.
Hepatocytes release metabolites which are toxic to cocultured brain cells
Two chemicals were compared: Cylophosphamide (CP), known to be converted in liver to a metabolite which is cytotoxic for extrahepatic tissues but without a specific brain toxicity, and isofenphos (Bayer AG), a re-presentative organophosphate known to be neurotoxic in humans. Brain cells were exposed directly, in cocultures or with the supernatant collected from exposed hepatocytes (exposure time 6 h – 24 hours). Cultured chicken brain cells died only after CP had been in contact with hepatocytes (supernatant or cocultures) (Fig.3). This proves that the metabolic competence of the hepatocytes is preserved and comparable to the situation in the living organism.
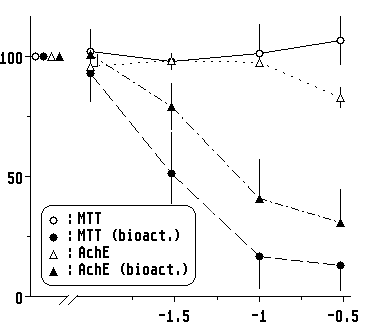 |
| Fig. 3. Dose response curves with Cyclophosphamide. Brain cells are exposed directly ( |
Brain cell specific functions can be impaired without cell death
Isofenphos, in the dose range chosen, inhibited AchE activity without affecting cell viability, again only after contact with hepatocytes (Fig.4). This inhibition of an organ-specific function (in this case the cholinergic nerves) prior to an overall cytotoxicity contrasts with the behaviour of CP (3) and corresponds well with a tissue-specific neurotoxic activity.
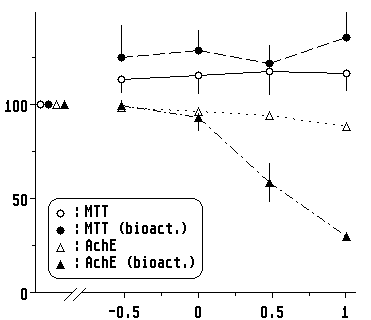 |
| Fig. 4. Dose response curve with isofenphos. Brain cells show an inhibition of AchE only after exposure to the supernatant ( |
Potential of cocultures
The addition of hepatocytes to cultures of chicken brain cells provides a metabolic activation system comparable to that of the liver. Testing the cytotoxicity of the supernatant from hepatocytes treated with the parent compounds provides a method to measure the stability and half-life of the reactive metabolites under investigation.
The present experimental model allows the discrimination between the induction of general cytotoxicity (MTT activity) and specific neurotoxic activity (inhibition of AChE activity without cytotoxicity) by chemicals and drugs.
Combinations of hepatocytes with cell cultures from other extrahepatic tissues, or co-cultures using different cell types from the same tissue (e.g. nerve cells and glial cells or hepatocytes and Kupffer cells (4)) allow intercellular interactions to be identified (including intercellular signalling), which take place within the intact organism and might affect cellular toxicity or the pharmacological behaviour of drugs.
Individual cell populations can be pretreated with modulators of xenobiotic metabolism before combining in cocultures. Such an approach enables the investigation of specific aspects of pharmacological pathways.
Co-cultures in the broad sense have not only the potential to provide a test systems which mimic more closely the situation in the living organism, but can also contribute to the elucidation of interactions which might be responsible for divergent results in toxicity and pharmacological tests in vitro versus in vivo.
Published updated version of this Bulletin 13/2007 (PDF)
References:
- Maier P., Saad B. and Schawalder H.P. (1994) Effect of periportal- and centrilobular-equivalent oxygen tension on liver specific functions in long-term rat hepatocyte cultures. Toxic. in Vitro 8, 423-435.
- Bruinink A. (1992) Serum-free monolayer cultures of embryonic chick brain and retina: Immunoassays of developmental markers, mathematical data analysis and establishment of optimal culture conditions. In: The Brain in Bits and Pieces (Ed. G. Zbinden) pp. 23-50, MTC Verlag, Zollikon.
- Bruinink A., Yu D. and Maier P. (2000) Short term assay for liver cell activated neurotoxic drugs. submitted.
- Milosevic N., Schawalder H.P. and Maier P. (1999) Kupffer cell mediated differential down-regulation of cytochrome P450 me-tabolism in rat hepatocytes. Europ. J. Pharmacol. 368,75-87.
| [*] | The use of human cellsGenetic differences between human and laboratory animals (in most cases rodents) introduce an uncertainity when extrapolating neurotoxicity data from animal experiments to humans. One approach to overcome this problem is the comparison of results obtained with primary cultured human central nervous systems cells to those from animal cell cultures. However this approach is limited for ethical and legal reasons by the availability of human tissue sources. Primary human (as well as animal) brain cells have to be cultured from fetal tissue obtained from therapeutic abortions. This is because brain cells usually survive only when seeded in an immature state, with an optional culture period for differentiation into the different brain cells (neurons, glial cells, astrocytes or oligodendrocytes). Primary human cultures therefore, although the ideal in vitro system, cannot be used for routine testing. |
| Letzte Änderung: 30.01.2008 |