 |
de | fr | en print view![]()
Annual report 2017
The 3R Research Foundation in 2017
In 2017 the Foundation provided a total of Fr. 304,156.80 for 11 research projects that had been approved previously. The Administrative Board received the final reports for four completed projects. No new applications were approved owing to the fact that the Foundation now has funding only for ongoing projects. The Foundation will receive no more funding from the federal authorities or Interpharma.
Consequently the Foundation's activities were limited mainly to transferring funds to ongoing projects.
3R-Principles
The 3Rs are Replace, Reduce and Refine animal experimentation. The 3Rs must be the guiding principles behind animal experimentation; if a study can be carried out without using any laboratory animals then such a procedure must be used. If it is essential to use laboratory animals under the terms of animal protection legislation the number used must be kept to a strict minimum. The third “R” requires that animals used for laboratory experiments be made to suffer an absolute minimum of pain and/or stress. The 3R Research Foundation funds research projects whose aim is to improve present-day experimental methods from the point of view of the 3Rs.
Summary of the Year’s Activities
The Foundation’s website
Detailed information about all the Foundation’s activities can be found on its website at www.forschung3r.ch.
Eleven projects subsidised
A total amount of Fr. 304,156.80 was paid out for 11 ongoing projects during 2017.
Four projects successfully completed
In-vitro engineering of a human cell-based three-dimensional dynamic model of atherosclerosis (135-15) Dr. Benedikt Weber, Swiss Centre for Regenerative Medicine, University Hospital Zurich. The most common cause of death among people in the western hemisphere is cardiovascular disease. In the majority of cases, the causes of the disease can be traced back to lesions in the arterial cell walls, so-called atherosclerotic plaque. At present, various animal models are used to investigate the origin of cardiovascular disease as well as for developing and testing new medication aimed at preventing/arresting or even curing the disease.
In this project, the research team have succeeded in developing a three-dimensional cell culture system from human atherosclerotic plaque (from material obtained following heart and blood vessel surgery) whereby the variety of cells involved in the disease are included in the model. The system is also capable of imitating the pulsatile blood vessel phenomena for simulating the biomechanical forces that play an important role in the origin of cardiovascular disease. This system provides a new biological "tool" for this vast research field that will help to avoid the necessity of using a large number of laboratory animals.
A new in-vitro vascular model mimicking the endothelial barrier (139-14) Dr. Marietta Hermann, AO Research Institute, Davos. Blood vessel walls, which are lined with endothelial cells that in turn are covered with pericytes, control the transport of various substances (nutrients, gases such as oxygen, etc.) in both directions (from the blood to tissue cells and vice versa). Cells also pass through this wall, e.g. antibody cells, as they spread from the blood into tissue to eliminate invasive pathogens. Until now it has not been possible to simulate this barrier function in vitro. For this reason, many experiments involving laboratory animals (mice, rats, etc.) are still being carried out in order to assess the expected barrier effect on new active substances or the extent to which antibodies can penetrate the endothelial barrier.
The successful creation of a comprehensive in-vitro blood vessel model including pericytes would provide a new research tool that would obviate the need for many experiments involving laboratory animals. The aim of this project was to create such an in-vitro system (micro-fluid chamber, basal membrane system, pericytes, etc.). Unfortunately, the technical difficulties encountered by the research team proved so extreme that they were unable to achieve their aim. Nevertheless, by designing a new modified chamber model, they did succeed in solving some of their basic problems, for example, the inclusion of air bubbles and various points of leakage in the complicated set-up.
Hydrocephalus simulator for testing active ventriculoperitoneal shunts (140-14) Dr. Marianne Schmid Daners, Institute for Dynamic Systems and Control, ETH Zurich. The ventriculoperitoneal shunts that are used in medicine today to treat hydrocephalus were developed in the 1950s. They drain brain fluid that cannot drain naturally (or be recirculated) into the abdomen. Passive pressure valves are used in these shunts that often become blocked and consequently lead to various complications. New systems are normally tested on laboratory animals.
In order to avoid such testing on animals, a new platform has been developed that consists of a simulator whereby ventriculoperitoneal shunts can be tested in vitro. The laboratory run by the leader of this project plans to build such simulators independently at first. The simulators will then be made available to scientists working at universities.
In-vitro alternatives to in-vivo bioconcentraton-testing in fish: restricted to rainbow trout or broadly applicable? (141-14) Prof. Helmut Segner, Centre for Fish and Wildlife Health, Vetsuisse Faculty, University of Berne. Throughout the world there are programmes for regulating the use of chemicals such as pesticides and for estimating the risk of persistence, toxicity and accumulation, in particular bioaccumulation, in live animals. One of the most frequently used models for such evaluations concerns bioaccumulation in fish, namely in their liver cells. High numbers of laboratory fish are required for such tests. New alternative testing methods to in-vivo measurements in fish include in-vitro and in-silico methods using liver cell systems from rainbow trout, a cold-water fish.
In this project the research team succeeded in demonstrating that the data obtained from rainbow trout material (liver cells) could be reproduced using material from carp (which is often used in such tests in Asian countries). With this method it should be possible to replace animal testing totally or at least partially. The data obtained represent a major step towards the acceptance of an animal-free approach to determining the predictive bioaccumulation of chemicals in fish for the purposes of regulation in the future.
3R-Info-Bulletins
No new 3R-Info Bulletins were published in 2017.
3R-Info bulletins are published on the Foundation’s website (www.forschung3r.ch/en/publications/index.html).
1. Background of the Foundation
The Foundation is a cooperative institution set up by the Parliamentary Group for Animal Experimentation Questions (public organ), Interpharma (association of pharmaceutical companies that carry out research in Switzerland; www.interpharma.ch/thema/uberinterpharma) and the Animalfree Research Foundation (animal protection). The Foundation was entered in the commercial register on 18 August, 1987.
The funds for subsidising research are provided principally by the Federal Food Safety and Veterinary Office and Interpharma.
2. Purpose of the Foundation
The purpose of the 3R Research Foundation Switzerland is to promote alternative research methods through grants for research projects as well as to implement and promote the 3R principles. The organisation supports first and foremost projects aimed at developing new methods or refining accepted methods (validation) which offer improvements vis-à-vis standard animal experimentation in line with the 3R motto, Replace, Reduce, Refine.
A broad range of projects is funded on the condition that they are likely to replace animal experimentation or to reduce the number of animals used or the stress and/or pain suffered. Accordingly, projects based on the Foundation’s three principles and covering any of a broad selection of bio-medical disciplines will be taken into consideration.
In 2017 the Foundation approved no new projects, since it received funding from the federal authorities and Interpharma for the last time in 2016 for ensuring that ongoing projects could be completed. In the future, funding from these two sources will be made available to the new 3R Competence Centre that was founded in 2018 under the auspices of "swissuniversities". The 3R Research Foundation will therefore cease its activities and be disbanded in 2018 as soon as the ongoing projects have been completed.
3. Organisation of the Foundation
The Administrative Board
The Administrative Board of the Foundation is made up of nine members, two representing the Swiss parliament, two representing animal protection, two from Interpharma and two from the Federal Food Safety and Veterinary Office, as well as a representative of other interested circles. Current members are:
Joachim Eder, member of the Council of States, Unterägeri (Chairman)
Dr. Peter Bossard, Horw (Deputy Chairman)
Dr. Philippe Bugnon, Institute of Laboratory Animal Science, University of Zurich
Dr. Isabelle Chevalley, member of the National Council, St. George (as from 26.5.15)
Dr. Kaspar Jörger, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Ingrid Kohler, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Birgit Ledermann, Novartis Pharma Ltd, Basle
Claudia Mertens, biologist, Zurich Animal Protection League, Winterthur
Nathalie Stieger, economist, F. Hoffmann-La Roche Ltd., Basle
The Evaluation Committee
Prof. Ernst B. Hunziker, University Hospital, Berne (Chairman)
Prof. Hans Acha-Orbea, Department of Biochemistry, University of Lausanne
Dr. Franziska Boess, F. Hoffmann-La Roche Ltd, Basle
Dr. Urban Deutsch, Theodor-Kocher-Institute, University of Berne
Dr. Robert Friis, University of Berne
Prof. Andrew Hemphill, Institute of Parasitology, University of Berne
Dr. Ingrid Kohler, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Kurt Lingenhöhl, Novartis Pharma Ltd, Basle
Prof. Matthias Lutolf, Lausanne Federal Institute of Technology
Prof. Thomas Lutz, Institute of Veterinary Physiology, University of Zurich
Prof. Alex Odermatt, Department of Pharmaceutical Sciences, University of Basle
Prof. Tatiana Petrova, University of Lausanne
Prof. Barbara Rothen-Rutishauser, Adolphe Merkle Institute, University of Fribourg
Dr. Stefanie Schindler, Animalfree Research Foundation, Berne
Scientific adviser
Prof. Ernst B. Hunziker, University Hospital, Bern
Administrator
Ernst P. Diener, lawyer, Münsingen
Auditors
DieWirtschaftsprüfer.ch AG, Thun
Supervisory body
Federal Department of Home Affairs
Articles and statutes of the Foundation
- Deed of foundation dated 13 February 1987, modified on 28 September 2011
- Regulations dated 30 March 2011 1987 (latest update 4 December 2014)
- Guidelines for awarding research grants dated 15 May 1987 (latest update 4 December 2014)
4. Personnel
In 2017 there were no changes in the Administrative Board nor the Evaluation Committee.
5. Activities during 2017
In the Foundation’s thirty-first year of existence the Administrative Board met twice, namely in April and December, for a half-day meeting. Apart from the statutory business concerning the end of the business year 2016, the Board addressed the following issues.
In April, the Board focused on the financial statements for 2016 and earmarking research funds for ongoing projects. In addition, it took note of the final reports on three completed projects.
At the December meeting, the Administrative Board was given a progress report on the creation of the 3R Competence Centre and received the final report on one project. Since, in view of its current aims and activities there is no future for the 3R Research Foundation, the Administrative Board decided to dissolve the Foundation once its obligations in relation to research grants have been fulfilled. The existing funds should suffice to meet all current obligations. The meeting finished with a review of activities during 2017 and those planned for 2018, which was followed by way of a thank-you for the work carried out in 2017 by a dinner for the whole Board.
In view of the decision not to call for any further applications, the Evaluation Committee did not meet during 2017. The Scientific Advisor organised the review of the annual reports and final reports by mail. We should like to take this opportunity to thank the members of the Evaluation Committee for their voluntary work in this connection.
6. Overview of the number of applications and approvals
During 2017 four projects were completed (135-13, 139-14, 140-14, 141-14). Together with those projects completed earlier, this brings the total of finished projects to 138 out of 146.
7. Financial business
Research funding for the 11 ongoing projects amounted to Fr. 304,156.80 in 2017. The sum of Fr. 2,000.00 was used for participation in meetings where projects funded by the 3R Foundation were presented. Expenditure on project supervision and information (Fr. 12,650.00 + Fr. 2,662.40) totalled Fr. 15,312.40. Together with the balance of provisions for project funding (-Fr. 304,156.10 = Fr. 77,685.25 for provisions for 2018 minus Fr. 381,841.35 liquidation of provisions for 2017), and the provision of Fr. 8500.00 for project supervision in 2018, total expenditure for research projects amounted to Fr. 25,813.10. Administrative costs totalled Fr. 54,056.70. With a provision of Fr. 62,800.00 for administrative costs for 2018, total expenditure therefore amounted to Fr. 142,669.80.
On the income side, the equal financial commitment of the federal authorities and Interpharma has thus far constituted the basis for the Foundation's activities. The Foundation received no funding from the FSVO or from Interpharma in 2017, which meant that income was zero.
The balance therefore shows an excess of expenditure over income of Fr. 142,669.80. This amount appears as a withdrawal of funds against capital funds on the balance sheet. Consequently, capital funds or unused research funding fell from Fr. 236,235.52 at the end of 2016 to Fr. 93,565.72 at the end of 2017.
At the end of the year under review the sum earmarked by the Administrative Board on the basis of project approvals but as yet not paid out amounted to Fr. 77,685.25. This amount is fully covered by provisions. Consequently, as at 31.12.2017, there remained no contingency commitments that are not shown in the financial statements.
The budget for 2018 includes Fr. 77,685.25 for ongoing projects.
8. Overview of grants awarded between 1987 and 2017
Together the federal authorities and Interpharma have contributed Fr. 23,826,000.00 to the Foundation since 1987. At the end of 2017 a total of Fr. 19,632,951.45 had been granted for projects and other subsidies, of which Fr. 19,555,266.90 had been paid out so far. Expenditure for project evaluation and supervision amounted to Fr. 2,329,800.08 and the accumulated administrative costs totalled Fr. 2,018,566.37 (8.6% of total expenditure or 10.4% of grants paid).
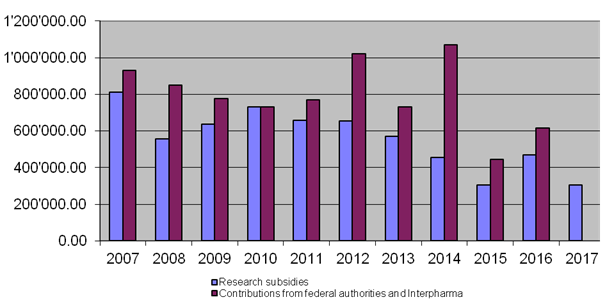
10-year overview
9. Annual financial statements
| Profit and loss account | 2017 | 2016 |
|---|---|---|
| Federal contribution | 0.00 | 365 000.00 |
| Interpharma contribution | 0.00 | 250 000.00 |
| Contributions to the Foundation | 0.00 | 615 000.00 |
| Research grants | -306 156.80 | -472 172.90 |
| Reimboursement of research grants | 0.00 | 0.00 |
| Adjustment reserves for research grants | 304 156.10 | 205 481.45 |
| Project supervision and information | -15 312.40 | -77 758.60 |
| Project supervision and information | -8 500.00 | |
| Balance for current projects | -25 813.10 | 270 549.95 |
| Administrative costs | -54 056.70 | -59 637.20 |
| Intermediate balance | -142 669.80 | 210 912.75 |
| Financial income | 0.00 | 61.60 |
| Financial result | 0.00 | 61.60 |
| Allocation to capital funds | 0.00 | -210 974.35 |
| Withdrawal from capital funds | 142 669.80 | |
| Balance | 0.00 | 0.00 |
| Balance as per 31st December | 2017 | 2016 |
| Assets | ||
| Liquid Assets | 249 270.97 | 620 632.57 |
| Accounting apportionment assets | 0.00 | 1 778.80 |
| Current assets | 249 270.97 | 622 411.37 |
Liabilities | ||
| Accounting apportionment liabilities | 5 720.00 | 3 334.50 |
| Reserves for research grants | 148 985.25 | 381 841.35 |
| Borrowed capital | 154 705.25 | 385 175.85 |
| Capital | ||
| – Carried forward 1 January | 236 235.52 | 25 261.17 |
| – Change in capital | -142 669.80 | 210 974.35 |
| Balance as at 31 December | 93 565.72 | 236 235.52 |
| Foundation's capital | 1 000.00 | 1 000.00 |
| Organisational capital | 94 565.72 | 237 235.52 |
| 249 270.97 | 622 411.37 | |
Contingent liabilities
Keine
Münsingen, 23 February 2018
3R RESEARCH FOUNDATION
The Chairman: signed Joachim Eder
The Administrator: signed Ernst P. Diener
10. Auditors' report to the Administrative Board
DieWirtschaftsprüfer.ch AG in Thun audited the financial statements for the year according to standards of limited auditing and did not find any indication that the accounts and statements do not correspond to current legislation or the principles and regulations of the Foundation.
| Last modified 2018/06/29 |