 |
de | fr | en print view![]()
Annual report 2014
The 3R Research Foundation in 2014
In 2014 the Foundation provided a total of Fr. 401,912.85 for 14 research projects that had been approved previously. Eight projects were successfully completed and the Administrative Board approved 4 new projects. The latter, with a high level of relevance to the Foundation's 3R principles, were selected by the Evaluation Committee from the 45 project outlines originally submitted and were recommended for research grants. 3R Info Bulletins 52 and 53 included the results of two completed projects. The Confederation and Interpharma together provided Fr. 1,070,000, a higher amount than usual. This means that research grants for 2014 and reserves for research grants for 2015 are covered.
In June a delegation from the Danish 3R Centre met representatives of the Foundation to exchange information on their experience. The Danish 3R Centre is in the throes of being set up and the delegation was interested to hear about our experience over the years. In September the Administrative Board was informed of the results of a study into the extent to which the Foundation's research grants achieved its aim. These results showed that new methods tended to be applied principally in the project leader's immediate area, and that typical keywords should be used in publications so that it is easier to find the papers in the scientific databases.
In December elections were held for the Administrative Board and the Evaluation Committee for the period 2015-2018. Following sweeping changes in 2013, the existing Administrative Board was re-elected in toto for a further 4-year period. Three new experts were elected to the Evaluation Committee.
3R-Principles
The 3Rs are Replace, Reduce and Refine animal experimentation. The 3Rs must be the guiding principles behind animal experimentation; if a study can be carried out without using any laboratory animals then such a procedure must be used. If it is essential to use laboratory animals under the terms of animal protection legislation the number used must be kept to a strict minimum. The third “R” requires that animals used for laboratory experiments be made to suffer an absolute minimum of pain and/or stress. The 3R Research Foundation funds research projects whose aim is to improve present-day experimental methods from the point of view of the 3Rs.
Summary of the Year’s Activities
The Foundation’s website
Detailed information about all the Foundation’s activities can be found on its website at www.forschung3r.ch.
14 projects subsidised
A total amount of Fr. 401,912.85 was paid out for 14 ongoing projects during 2014.
Four new projects
Four new projects were approved in 2014 for which a total of Fr. 435,665.00 has been earmarked. These new projects are described in detail in the list of funded projects on the Foundation’s website (www.forschung3r.ch/en/projects/index.html).
Validation of human stem-cell pluripotency using a bioreactor-based culturing system instead of a murine model to effect the development of embryoid bodies into teratomas (142-14) Prof. Christian de Geyter, Department of Biomedicine, Basle University Hospital, Basle, Switzerland. After stem cells from donor tissue have been isolated (or cultured) it must be checked whether they have maintained their typical capacity to differentiate into various types of tissue (pluripotency). According to international guidelines, such tests for pluripotency of stem cells are normally carried out using mice with a non-functional immune system. The authors propose to develop a new bioreactor-based system which will enable researchers to examine the differentiation of stem cells into various types of tissue in vitro.
In-vitro alternatives to in vivo bioconcentration-testing in fish: restricted to rainbow trout or broadly applicable? (141-14) Prof. Helmut Segner, Centre for Fish and Wildlife Health, University of Berne, Switzerland. Xenobiotic substances are chemical compounds that accumulate as foreign substances (e.g. toxins) in organisms where they are normally not found. According to OECD Guidelines (TG 305, Guidelines for Testing of Chemicals, Degradation and Accumulation), the potential risks of such substances must be tested on animals. Many animals are used for such tests. The project leader has already developed a liver cell culture system using cells from trout, a cold-water fish, whereby the testing can be carried out in vitro (Project 108-07). The researchers now propose to adapt this system for liver cell cultures from carp, a warm-water fish, in order to enable the in vitro testing to be carried out in various other parts of the world.
Hydrocephalus simulator for testing of active ventriculoperitoneal shunts (140-14) Dr. Marianne Schmid Daners, Institute for Dynamic Systems and Control, Department of Mechanical and Process Engineering, Zurich Federal Institute of Technology, Zurich, Switzerland. The present generation of valves and shunts used for treating hydrocephalus is out-of-date. Blockages often occur in the artificial drainage system for transferring excess cerebral fluid from the brain to the abdominal cavity. As a rule, the reliability of new products or systems is tested on larger animals. In order to alleviate the need for such animal testing, a new platform will be developed consisting of a simulator which can be used for testing ventriculoperitoneal shunts in vitro.
A new in-vitro microvascular model of the endothelial barrier (139-14) Dr. Marietta Herrmann, AO Research Institute Davos, Switzerland. The walls of the blood vessels that are lined with endothelial cells, which in turn are covered with pericytes, control the transfer of substances (nutrients, gases such as oxygen, etc.) in both directions (from the blood to the tissue cells and vice versa). Cells also pass through this barrier, e.g. antibody producing cells, and access the tissue from the blood to eliminate pathogenic organisms that have entered the system. As a rule, studies of the mechanisms whereby substances and cells pass through the barrier involve the use of laboratory animals, mainly transgenic mice. The aim of this research project is to develop a new in vitro culture system for the blood vessel wall whereby such studies can be carried out in vitro.
Eight projects successfully completed
Organotypic brain-slice cultures derived from brains obtained from slaughterhouses as an in-vitro alternative for the investigation of neuroinfectious diseases in ruminants (116-09) Prof. Anna Oevermann, Vetsuisse Faculty, University of Berne, Switzerland. Infections of the brain or of the central nervous system, for example through bacteria (Listeria), viruses (BSE) or active protein molecules (prions) normally lead to serious disease in humans or animals. So far, there is a lack of laboratory models, for example in vitro simulation, for studying such diseases. Prof. Oevermann has succeeded in developing a culture model for investigations in this area by using nerve tissue taken from animals obtained from slaughterhouses.
Development of an in-vitro model from embryonic stem cells for identifying tissue inflammation as a reaction to implanted material (INFPLANT) (117-09) Prof. Maria Wartenberg, Department of Molecular Cardiology, University Clinic Jena, Germany. The development of new implant materials for artificial hip joints, etc. involves, among other things, testing the rate of tolerance among recipients. Normally this is done using live animals. Prof. Wartenberg has succeeded in developing a tissue tolerance test using embryonic stem cells. This in vitro method provides valuable information as to potential tolerance in humans.
Nerve-cell mimicking liposomes as an in-vitro alternative to potency-testing of toxins with multi-step pathways, such as Botolinum neurotoxins (125-11) Dr. Oliver G. Weingart, Institute for Food, Nutrition and Health, Zurich Federal Institute of Technology, Switzerland. Botulinum toxins are not only extremely dangerous substances that are produced by bacteria in cases of infection and can lead to nerve paralysis, but are also used in a cosmetic preparation to eliminate wrinkles caused be ageing. Normally such, neurotoxins are tested on laboratory animals. In this project, Dr. Weingart has achieved a major step towards developing a new in vitro based efficacy test for these substances.
Model development and validation to investigate myeloid-cell homeostasis (126-11) Dr. Charaf Benarafa, Theodor Kocher Institute, University of Berne, Switzerland. The blood’s defence cells live for only a short time – a matter of hours – and for this reason many animals are required for research in this field. In order to replace these animals in the future, Dr. Benarafa and his team attempted to make such cells "immortal" so that many fewer or even no animals would need to be sacrificed. Unfortunately it was found that cells transformed in this way lose an important defence ability and certain other characteristics with the result that they are no longer of use in research.
Genetic modification of the human airway epithelium – a paradigmatic system to study host responses to human respiratory viruses (128-11) Prof. Volker Thiel, Institute of Immunobiology, Cantonal Hospital St. Gallen, Switzerland. Many infectious diseases in humans start off in the airways, where germs manage to adhere to the airway epithelium and infect the victim’s body (colds, influenza, etc.). Research in this field is based principally on animal experimentation. Prof. Thiel has succeeded in devising an in-vitro epithelium cell model which will enable researchers to study the development of such human respiratory diseases in vitro.
Using a microfluidic chamber to study mitochondrial transport in PTEN and SOCS3 dependent axonal regeneration (129-11) Prof. Zhigang He, Children's Hospital, Boston, USA. Research into healing processes in damaged nerve fibers (axons) and testing of new substances for promoting such healing rely heavily on the use of laboratory animals. Prof. He and his team at the Boston Children's Hospital in the USA have succeeded in developing a new in-vitro system for investigating the early processes in the healing of such fibers using cell cultures.
Antibody-phage-selection strategy for application in non-specialized laboratories (131-11) Prof. Christian Heinis, Therapeutic Peptides and Proteins Laboratory, EPFL Lausanne Federal Institute of Technology, Switzerland. Most antibodies used in research are still produced by immunizing animals. Prof. Heinis and his team succeeded in the development of an antibody scFv phage display library that may be distributed to laboratories free of charge and without any intellectual property (IP) constraints. From this library, antibodies to targets of choice can be isolated in vitro, omitting standard techniques based on animal immunization. In addition, they developed a phage display selection strategy with significantly fewer experimental steps that should facilitate the in vitro generation of affinity ligands by non-experts. The proposed method should replace animal experiments that are commonly performed to develop polyclonal and monoclonal antibodies.
Identification of predictive in-vitro markers for hematopoietic stem cell function (133-12) Prof. Matthias P. Lutolf, Laboratory of Stem Cell Bioengineering, Institute of Bioengineering, EPFL Lausanne Federal Institute of Technology, Switzerland. Human hematopoietic stem cells have been successfully used in medicine to treat leukaemia. Before the treatment can be given, many tests must be carried out to determine the stem cell characteristics and various other functions and abilities of the cells, which normally involves laboratory animals. Prof. Lutolf has succeeded in developing a new in-vitro method for obtaining this information whereby some of the tests can be done in vitro using new markers.
3R-Info-Bulletins
3R-Info bulletins are published on the Foundation’s website (www.forschung3r.ch/en/publications/index.html).
Genetic manipulation of cultured human airway epithelium – a paradigmatic system to study host responses to human respiratory viruses (no. 53, November 2014) The airway epithelium is the main port of entry for many respiratory pathogens and an important barrier to infection. Experimental systems that are suitable for studying basic virus-host interactions are scarce and are still preferentially performed in animal models. Prof. Thiel and his team succeeded to establish a unique in-vitro airway epithelial-cell culturing system that permits the molecular analysis of host-pathogen interactions at the port of entry of many respiratory pathogens.
A new in-vitro approach to the study of brain tumours: an alternative to in-vivo experiments in animals (no. 52, June 2014) In humans, glioblastomas are the most common and most aggressive type of brain tumours. Animal experimentation as part of research into glioblastomas causes extreme suffering since it involves implanting a tumour in the brain of a mouse. Together with his research team at the University of Geneva, Dr. Olivier Preynat-Seauve has succeeded in developing a cell culture model whereby the interaction between the tumour cells and nerve tissue can be simulated. Through this model, experiments that cause considerable suffering to the mice should become superfluous.
1. Background of the Foundation
The Foundation is a cooperative institution set up by the Parliamentary Group for Animal Experimentation Questions (public organ), Interpharma (association of pharmaceutical companies that carry out research in Switzerland; www.interpharma.ch/thema/uberinterpharma) and the Animalfree Research Foundation (animal protection). The Foundation was entered in the commercial register on 18 August, 1987.
The funds for subsidising research are provided principally by the Federal Food Safety and Veterinary Office and Interpharma.
2. Purpose of the Foundation
The purpose of the 3R Research Foundation Switzerland is to promote alternative research methods through grants for research projects as well as to implement and promote the 3R principles. The organisation supports first and foremost projects aimed at developing new methods or refining accepted methods (validation) which offer improvements vis-à-vis standard animal experimentation in line with the 3R motto, Replace, Reduce, Refine.
A broad range of projects is funded on the condition that they are likely to replace animal experimentation or to reduce the number of animals used or the stress and/or pain suffered. Accordingly, projects based on the Foundation’s three principles and covering any of a broad selection of bio-medical disciplines will be taken into consideration.
3. Organisation of the Foundation
The Administrative Board
The Administrative Board of the Foundation is made up of nine members, two representing the Swiss parliament, two representing animal protection, two from Interpharma and two from the Federal Food Safety and Veterinary Office, as well as a representative of other interested circles. Current members are:
Joachim Eder, member of the Council of States, Unterägeri (Chairman)
Dr. Peter Bossard, Horw (Deputy Chairman)
Dr. Philippe Bugnon, Institute of Laboratory Animal Science, University of Zurich
Dr. Isabelle Chevalley, member of the National Council, St. George (as from 26.5.15)
Dr. Kaspar Jörger, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Ingrid Kohler, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Birgit Ledermann, Novartis Pharma Ltd, Basle
Claudia Mertens, biologist, Winterthur
Nathalie Stieger, economist, F. Hoffmann-La Roche Ltd., Basle
The Evaluation Committee
Prof. Ernst B. Hunziker, University of Berne (Chairman)
Prof. Hans Acha-Orbea, Department of Biochemistry, University of Lausanne
Dr. Franziska Boess, F. Hoffmann-La Roche Ltd, Basle
Dr. Urban Deutsch, Theodor-Kocher-Institute, University of Berne
Dr. Robert Friis, University of Berne (as from 1.1.15)
Prof. Marianne Geiser Kamber, Institute of Anatomy, University of Berne (up to 31.12.14)
Prof. Andrew Hemphill, Institute of Parasitology, University of Berne
Prof. Simon P. Hoerstrup, Swiss Centre for Regenerative Medicine, University Hospital Zurich (up to 31.12.14)
Dr. Ingrid Kohler, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Kurt Lingenhöhl, Novartis Pharma Ltd, Basle
Prof. Matthias Lutolf, Lausanne Federal Institute of Technology (as from 1.1.15)
Prof. Thomas Lutz, Institute of Veterinary Physiology, University of Zurich
Prof. Alex Odermatt, Department of Pharmaceutical Sciences, University of Basle
Prof. Tatiana Petrova, University of Lausanne (as from 1.1.15)
Dr. Stefanie Schindler, Animalfree Research Foundation, Berne
Scientific adviser
Prof. Ernst B. Hunziker, University of Berne
Administrator
Ernst P. Diener, lawyer, Münsingen
Auditors
Waber Treuhand GmbH, Spiez
Supervisory body
Federal Department of Home Affairs
Articles and statutes of the Foundation
- Deed of foundation dated 13 February 1987, modified on 28 September 2011
- Regulations dated 30 March 2011 1987 (latest update 4 December 2014)
- Guidelines for awarding research grants dated 15 May 1987 (latest update 4 December 2014)
4. Personnel
All the current members of the Administrative Board were re-elected for a further period of office.
Prof. Geiser Kamber and Prof. Hoerstrup did not stand for re-election to the Evaluation Committee. Prof. Robert R. Friis from the University of Berne, Prof. Matthias Lutolf from the Lausanne Federal Institute of Technology and Prof. Tatiana Petrova from the University of Lausanne were elected to the Evaluation Committee for the period 2015-2018.
5. Activities during 2014
In the Foundation’s twenty-eighth year of existence the Administrative Board met three times, namely in May, September and December, for a half-day meeting. Apart from the statutory business concerning the end of the business year 2013, the Board addressed the following issues.
In May, the Board focused on the financial statements for 2013 and earmarking research funds for ongoing projects. In addition, it took note of the final reports on seven completed projects. The Board also appointed representatives for a meeting with a delegation from the Danish 3R Centre to discuss experience on both sides.
At its meeting in September, the focus was on funding for new projects. Out of a total of 8 project proposals selected by the Evaluation Committe from the 45 applications received, 4 were finally approved. A prognosis of the re-election or election of members of the Foundation's organs in December promised a high rate of continuity. The President informed the Foundation of his invitation to give a talk on questions of strategy at an animal experimentation meeting organised by the Swiss Animal Protection League. He declined to accept the invitation since, in his opinion, the timing was premature as long as the Federal Council's report on the demand tabled by the National Council's Committee for Science, Education and Culture concerning the 3R Research Foundation had not been published. In the meantime, the Administrative Board will appoint a delegation to have discussions with the Animal Protection League. Following discussion of the assessment study on research promotion, it was decided that project leaders should expressly agree to mention the relationship of their study to the 3R principles with the keywords "animal use alternatives" or "animal testing alternatives" in the publication that they are expected to produce in order to make it easier for other scientists to locate 3R publications.
At the Administrative Board’s December meeting, the current members of the Administrative Board and the Evaluation Committee were re-elected for the period 2015-2018. In addition, three new members were elected to the Evaluation Committee in order to extend the Committee's scientific expertise to cover areas of research that are highly relevant to the 3R principles and which until now have been poorly covered or not covered at all, as well as to avoid further increasing the work-load of the individual members. The project leaders' undertaking to use certain keywords in their publications was included in the revised guidelines for awarding research grants. Furthermore, the Administrative Board received another final project report. The meeting finished with a review of activities in 2014 and prospects for 2015, followed by a convivial dinner.
As always the Administrator dealt with all matters concerning the Foundation that cannot be passed on to anyone else. In particular, this includes preparing all the necessary information for the Administrative Board to take their decisions, as well as dealing with correspondence with applicants and project managers. He also dealt with payments, the book-keeping, closing the books at the end of the financial year and the budget. In addition, he prepares the Annual Report as well as texts for the Foundation’s website.
Under the chairmanship of the Scientific Advisor, the Evaluation Committee held two meetings during the year, where in particular they examined 45 new applications for funding of which they chose 8 likely projects; finally 4 were approved. In addition they examined the final reports on a total of eight completed projects and submitted them to the Administrative Board. We should like to take this opportunity to thank the members of the Evaluation Committee for their voluntary work in this connection.
The Scientific Advisor was mainly occupied with preparing the meetings of the Evaluation Committee, publishing the 3R Info Bulletins (on the Foundation’s website at www.forschung3r.ch), writing the brief scientific reports in English which present the projects receiving funding on the Foundation’s website and regularly updating these reports.
6. Overview of the number of applications and approvals
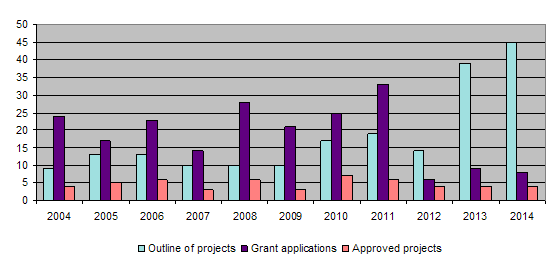
During 2014 eight projects were completed (116-09, 117-09, 125-11, 126-11, 128-11, 129-11, 131-12, 132-12). Together with those projects completed earlier, this brings the total of finished projects to 128 out of 142.
The bar-chart shows that the trend in the number of project outlines, detailed proposals and project approvals remained similar to the previous year. The numbers of project outlines and detailed proposals for 2013 and 2014 as depicted in the bar-chart cannot be compared with those of previous years since a new two-level application procedure was introduced in 2013. First, applicants submit project outlines which are assessed by the Evaluation Committee in the first round. Only those applicants putting forward a project with clear relevance to the Foundation's 3R principles are subsequently invited to submit a detailed project proposal.
Forty-five project outlines were submitted. After examining them all, the Evaluation Committee asked 8 applicants to submit a detailed proposal, and out of the 8 detailed proposals the Administrative Board finally approved 4 for a research grant. Regardless of the many applications received, there is little fluctuation in the number of projects approved for funding owing to the limited means available to the Foundation.
7. Financial business
The format of the profit and loss account for the Foundation has been revised to comply with the new law on financial reporting. For the fist time the budgeted research grants for the coming 12 months (Fr. 399,783.90) have now been booked as expenditure items for short-term reserves, and project supervision and information are included under project promotion. As a consequence, expenditure on project promotion, including research grants, reserves for contingent liabilities and project supervision, amounted to an extraordinary amount of Fr. 949,218.80. The balance of the profit and loss account is no longer shown as profit or loss, but as an allocation to or a withdrawal from capital funds, and is subsequently zero.
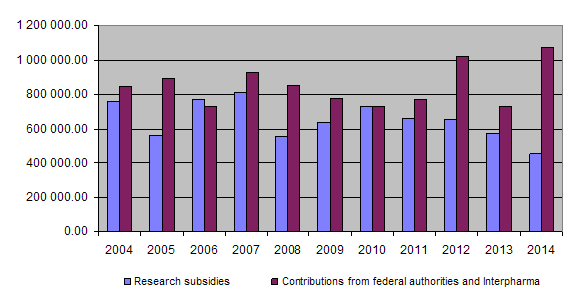
The equal financial commitment of the Confederation and Interpharma forms the basis of the Foundation's activities. In 2014 the FSVO (Federal Food Safety and Veterinary Office) provided the foundation with Fr. 535,000 while Interpharma transferred the sum of Fr. 365,000. The outstanding amount of Fr. 170,000 required to bring the latter's contribution up to the Confederation's level has been apportioned between accounting periods and booked as a debt under the Foundation's assets, on the basis of Interpharma's funding commitment of up to Fr. 500,000.00 per year. This means that the accounts for 2014 show an annual contribution of Fr. 535,000 from Interpharma. These exceptionally high contributions to the Foundation enabled us for the first time to include commitments for research grants for the coming 12 months in our financial statements without giving rise to an excess of expenditure over income.
At the end of 2014 the sum earmarked by the administrative Board in principle on the basis of project approvals but as yet not paid out amounted to Fr. 714,553.30. Out of this, Fr. 399,783.90 are covered by reserves. Subsequently, as at 31.12.2014, there remained contingency commitments in the amount of Fr. 314,769.50 that are not shown in the financial statements. The Foundation's credit balance with Interpharma arising from its Commitment VI amounted to Fr. 2,382,000.00 at the end of 2014.
The budget for 2015 includes Fr. 375,887.10 for current projects and around Fr. 500,000 for new projects.
8. Overview of grants awarded between 1987 and 2014
Together the federal authorities and Interpharma have contributed Fr. 22,766.000.00 to the Foundation since 1987. At the end of 2014 a total of Fr. 19,186,766.00 had been granted for projects and other subsidies, of which Fr. 18,472,212.70 had been paid out so far. Expenditure for project supervision amounted to Fr. 2,158,907.01 and the accumulated administrative costs totalled Fr. 1,865,936.82 (8% of total expenditure or 10% of grants paid).
10-year overview
9. Annual financial statements
| Profit and loss account | 2014 | 2013 |
|---|---|---|
| Federal contribution | 535 000.00 | 365 000.00 |
| Interpharma contribution | 535 000.00 | 535 000.00 |
| Contributions to the Foundation | 1 070 000.00 | 730 000.00 |
| Research grants | -454 511.05 | -572 162.35 |
| Adjustment reserves for research grants | -399 783.90 | 0.00 |
| Project supervision and information | -94 923.85 | -89 014.10 |
| Balance for current projects | 120 781.20 | 68 823.55 |
| Administrative costs | -113 717.50 | -107 014.00 |
| Intermediate balance | 7 063.70 | -38 190.45 |
| Financial income | 179.83 | 375.15 |
| Financial result | 179.83 | 375.15 |
| Non-operational income | 0.00 | 1305.70 |
| Allocation to capital funds | -7 243.53 | |
| Withdrawal from capital funds | 36 509.60 | |
| Balance | 0.00 | 0.00 |
| Balance as per 31st December | 2014 | 2013 |
| Assets | ||
| Liquid Assets | 509 678.94 | 259 826.78 |
| Accounts payable | 274.63 | |
| Accounting apportionment assets | 171 019.30 | 1 542.75 |
| Current assets | 680 698.24 | 261 644.16 |
Liabilities | ||
| Accounting apportionment liabilities | 21 309.45 | 9 282.80 |
| Reserves for research grants | 399 783.90 | 0.00 |
| Borrowed capital | 421 093.35 | 9 282.80 |
| Capital | ||
| – Carried forward 1 January | 251 361.36 | 287 870.96 |
| – Change in capital | 7 243.53 | -36 509.60 |
| Balance as at 31 December | 258 604.89 | 251 361.36 |
| Foundation's capital | 1 000.00 | 1 000.00 |
| Organisational capital | 259 604.89 | 252 361.36 |
| 680 698.24 | 261 644.16 | |
Contingent liabilities
Approved research grants not yet paid out Fr.. 314,769.50.
Münsingen, 22 April 2015
3R RESEARCH FOUNDATION
The Chairman: signed Joachim Eder
The Administrator: signed Ernst P. Diener
10. Auditors' report to the Administrative Board
Waber Treuhand GmbH in Spiez audited the financial statements for the year according to standards of limited auditing and did not find any indication that the accounts and statements do not correspond to current legislation or the principles and regulations of the Foundation.
| Last modified 2015/06/24 |