 |
3R-INFO-BULLETIN 14
May 2000
The Authors
   |
Prof. Dr. Isabel Roditi* is head of the laboratory for Molecular Parasitology at the University of Berne and carried out the present project together with her collaborators Dr. E. Vassella and Dr. Jean-Yves Salomone.
current address:
*Institute of General Microbiology,
University of Bern
Baltzerstr. 4,
CH-3012 Bern,
Switzerland.
isabel.roditi@imb.unibe.ch
Editor
Peter Maier, Scientific Adviser of the 3R Research Foundation
Transgenic protozoa as an alternative to transgenic animals
Production of bioactive proteins
Biomedical research depends increasingly on the production of substantial quantities of pure, functionally active proteins. Bacterial expression systems can provide large amounts of recombinant proteins, but these are frequently inactive and/or insoluble. Proteins produced in mammalian cells are more likely to be bioactive, but the yields are much lower. As a result, there is an increasing tendency to use transgenic animals as “bioreactors” [*]. Results from our laboratory suggest that unicellular organisms (protozoa) can provide an attractive alternative system for the medium-scale production of biologically active recombinant proteins (Table 1).
Table I Advantages of the expression system using procyclic trypanosomes
|
Trypanosomes as bioreactors
African trypanosomes (e.g. Trypanosoma brucei) are unicellular parasites that can be transmitted among mammals by the tsetse fly. Procyclic culture forms (Fig. 1) are equivalent to the forms found in the midgut of the tsetse fly. They are not infectious for mammals; furthermore the attenuated line that we use in our laboratory (strain 427) is completely safe since it cannot be translated by tsetse flies to a new animal host.
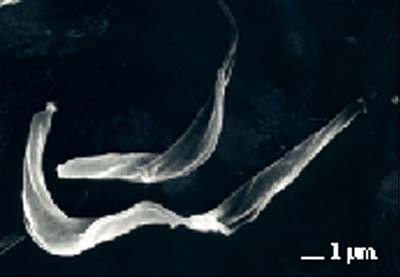 | |
| Fig.1. | Scanning electron micrograph of T. brucei procyclic forms. (micrograph kindly provided by Dr. A. Hemphill, University of Bern). |
Example 1: A vaccine candidate
More than half a million cattle die each year of East Coast Fever (ECF), a disease caused by tick-borne transmission of the protozoan parasite Theileria parva. Infection starts when sporozoite forms of the parasite enter lymphocytes. The sporozoite surface antigen p67 is the prime vaccine candidate against ECF. Sporozoites cannot be cultured and it is not possible to obtain them from infected ticks without allowing the ticks to feed on experimental animals (cattle and rabbits). Recombinant p67 that has been produced in either bacteria or baculoviruses cannot be recognised by antibodies. In the 3R research project we were able to create stable transformed trypanosomes containing different forms of p67. The most promising is a hybrid form of p67 that also contains sequences from an endogenous trypanosome surface protein (procyclin). Ongoing experiments will establish whether this form of p67 can substitute for the native Theileria p67 protein.
 | 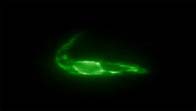 |
| Fig. 2a. Phase contrast picture of T. brucei. | Fig. 2b. Sublocalisation of p67 in the endoplasmic reticulum. (Immunofluorescence assay) |
Example 2: Lymphokines
Lymphokines are proteins that play an important role in the maturation and proliferation of cells of the immune system. They are normally produced in trace amounts in the bodies of humans and animals and can only be obtained in appreciable quantities by purification from tissues or alternatively by expression in transgenic organisms. Among other things, the lymphokine interleukin-4 (IL-4) can have a major influence on the host response to infection by bacteria or parasites.
Previous attempts to produce biologically active bovine interleukin 4 (IL-4) in bacterial and mammalian expression systems were unsuccessful. IL-4 produced by transgenic trypanosomes, however, is biologically active [3, 4].
Trypanosomes for drug testing
Two species of African trypanosomes can cause human sleeping sickness. Recent studies estimate 300 – 500 thousand new cases each year. There is an urgent need for new drugs to treat infected patients.
Bloodstream forms of the parasite survive and replicate in the blood of the mammalian host whereas procyclic forms - which normally first appear in the tsetse fly - can be lysed by serum. Compounds that can trigger premature differentiation of the parasite into procyclic forms in the bloodstream might therefore be of therapeutic value. We have produced transgenic bloodstream forms of trypanosomes that synthesise the bacterial enzyme b-glucuronidase (GUS) when they differentiate (for example, after exposure to a test compound) into procyclic forms [4]. This enzymatic activity can be detected by a colour change after adding a specific substrate to the cultures. This assay is so sensitive that it requires only minute quantities of trypanosomes easily cultured in vitro.
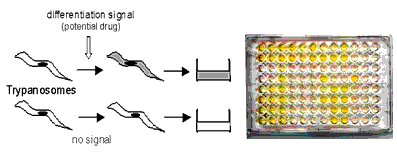 |
| Fig. 3. Principle of the GUS bio-assay. Each dark well in a micro-titerplate contains active GUS, showing that the test compound has triggered the trypanosomes to differentiate. |
References:
- Dobbelaere, D. A. E., Shapiro, S. Z. and Webster, P.(1985).Identification of a surface antigen on Theileria parva sporozoites by monoclonal antibody. Proc. Natl. Acad. Sci. USA, 82, 1771-1775.
- Furger, A., Schürch, N., Kurath, U. and Roditi, I. (1997). Elements in the 3’ untranslated region of procyclin mRNA regulate expression in insect forms of Trypanosoma brucei by modulating RNA stability and translation. Mol. Cell. Biol., 17, 4372-4380.
- Jungi, T.W., Brcic, M., Sager, H., Dobbelaere, D.A.E., Furger, A. and Roditi, I. (1997). Antagonistic effects of interleukin-4 and interferon-g on inducible nitric oxide synthase expression in bovine macrophages exposed to gram-positive bacteria. Clin. Exp. Immunol., 109, 431-438.
- Furger, A. Jungi, T. W., Weynants, V., Dobbelaere, D. and Roditi, I. Expression of biologically active bovine interleukin-4 in Trypansoma brucei. Manuscript in preparation.
- Sbicego, S., Vassella, E., Kurath, U., Blum, B and Roditi, I. (1999). The use of transgenic Trypanosoma brucei to identify compounds inducing the differentiation of bloodstream to procyclic forms. Mol. Biochem. Parasitol., 104, 311-322.
| [*] | Transgenic bacteria, protozoa, isolated cells and animals: “Transgenic” or “recombinant” organisms contain foreign DNA that is derived from another organism. The translation of the altered DNA sequence into the corresponding protein requires a complex multistep process (expression system). Bacteria have proven very useful for the production of large amounts of certain proteins (e.g. human insulin). In many cases, however, bacterial recombinant proteins are not active because bacteria cannot carry out important steps which occur normally in human or animal cells, such as attaching sugars to the proteins. Transgenic human or animal cells in tissue culture release recombinant proteins which often have the desired activity. However, the quantities of proteins are usually small and the foreign DNA may be unstable. Transgenic animals are derived from embryos or embryonic stem cells. The embryos or cells are microinjected with foreign DNA and then implanted into a recipient, pseudopregnant female. After birth, the animals are analysed for the presence of the new gene. The foreign gene can be tailored in such a way that large amounts of protein are produced (“bioreactor”) in specific tissues or in the milk or blood in order to recover the protein without sacrificing the animal. Animals in which specific genes (“knockouts”) are removed or replaced with an altered version serve different purposes. They are often used as models for human diseases. Protozoa (e.g. trypanosomes), as presented in this bulletin, are much closer to animals and humans on the evolutionary tree than are bacteria . They combine the advantages of bacteria (easy of handling) with those of mammals (bioactive protein, Table1). |
| Dernières modifications: 30.01.2008 |