 |
de | fr | en Druckansicht ![]()
3R-Project 125-11
Nerve-cell mimicking liposomes as in vitro alternative to potency-testing of toxins with multistep pathways, such as Botulinum neurotoxins
Oliver G. Weingart1, Marc-André Avondet2, Andreas Rummel3, Frank Gessler4, Peter Walde5, Martin J. Loessner1
1 Laboratory of Food Microbiology, Institute for Food, Nutrition and Health (IFNH), ETH Zurich, Schmelzbergstrasse 7, 8092 Zurich, Switzerland
2 Toxinology Group, SPIEZ LABORATORY, 3700 Spiez, Switzerland
3 Institut für Toxikologie, OE 5340, Medizinische Hochschule Hannover, Carl-Neuberg-Strasse 1, 30625 Hannover, Germany
4 miprolab GmbH, Marie-Curie-Str. 7, 37079 Göttingen, Germany
5 Federal Institute of Technology, Department of Materials, Wolfgang-Pauli-Strasse 10, 8093 Zürich, Switzerland
oliver.weingart@ilw.agrl.ethz.ch
Keywords: mice; toxicology; molecular biology: receptors; replacement; toxicity testing: neurotoxins; toxicity testing: pharmaceuticals; toxicity testing: receptor mediated
Duration: 2 years Project Completion: 2013
Background and Aim
Botulinum neurotoxins (BoNT), produced by the Gram-positive bacterium Clostridium botulinum, are the most lethal toxins known to man. Similar to other toxins with multiple active sites, BoNT exerts a complex function in the human body. In the latter case, the toxin binds via its binding domain (HCC = heavy chain binding domain) to specific cell-surface receptors on motoneurons. The translocation domain (HCN = heavy chain translocation domain) facilitates translocation of the endoproteinase domain (LC = light chain) into the nerve cell lumen, where the latter specifically cleaves distinct SNARE proteins. As a result, neurotransmitter release is inhibited and the adjacent muscle cells are paralysed. If untreated, this leads to cumulative paralysis, and eventually death by respiratory arrest. If applied in minute doses, however, the toxin exerts a locally constrained paralysing effect, which is employed in medical applications for the treatment of a wide range of diseases, disorders and in aesthetic surgery.
In the case of BoNT, the mouse LD50 test is so far the preferred method for batch control of BoNT-containing pharmaceutical products, such as BOTOX, Myobloc and others [1,2]. Hence, with an increasing number of medical applications, more than half a million mice are used each year for routine potency testing [3,4]. Owing to the often complex nature of multi-domain toxins such as BoNT, most existing cell-free in vitro methods fail to detect the entire toxic activity, instead measuring either merely part of their toxicity or only the toxins present, e.g. by immunological methods. Although cell culture assays have been established for use in toxicity testing, many of these assays still require expensive and difficult maintenance, qualified personnel and the appropriate cell culture facilities. In addition, if stem-cell derived cells are used, long differentiation times of up to four weeks can be necessary [5].
Here, we describe the groundwork for an in vitro alternative for testing the entire toxic activity of toxins with multiple active sites, taking BoNT type B (BoNT/B) as an example, using liposomes with integrated nerve cell receptors to imitate the motoneuron membrane. BoNT/B binds via its HCC to the presented receptors on the liposome surface. If the pH in the surrounding medium is lowered, the HCN translocates the LC into the liposomal lumen, where the latter exerts its endoproteinase activity by cleaving a peptide substrate with specific cleavage sites and a quenched fluorophore pair. Upon cleavage, the fluorophore pair is unquenched and its fluorescent signal can be detected, allowing for quantification of the actual potency of BoNT in the system. With the use of well defined components, the liposomes may be produced under reproducible conditions. Moreover, liposomes can be stored long term at little expense and reconstituted on demand [6,7]. Furthermore, if the respective binding molecules (i.e. receptors) and substrates are available, the modular character of the presented system may also allow for testing of other toxins with multistep pathways which exert complex (chain reactions) functional changes, which are difficult to measure, in cells and the body.
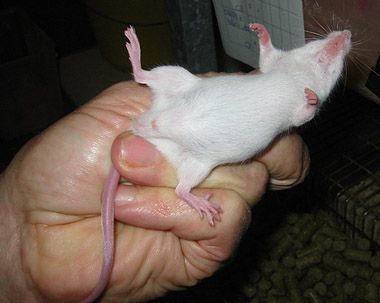
Figure 1: Mouse LD50 test: For batch control of BoNT containing pharmaceutical products, different dilutions of the product are injected intraperitoneally into mice. The mice are observed for typical botulism symptoms. If the dose is high enough, mice die of suffocation within 96 hours.
Figure 2
BoNT toxic activity and its effect at motoneurons (panels above) and the model of detection, usingfunctionalised liposomes (panels below).
In the Motoneuron: BoNT (in red and blue) is bound to the motoneuron via specific receptors integrated into its membrane. Subsequently, the toxin is passively taken up via endocytosis and becomes encapsulated into an endosome. During endosome-lysosome transition the interior of this vesicle becomes acidified. The drop in pH triggers the active transmembrane translocation of the toxin’s light chain (blue) via its heavy chain (red). Once in the cytosol, the light chain specifically cleaves SNARE-proteins. This leads to an inhibition of acetylcholine secretion. The following muscle cell becomes paralysed.
In the liposomes: Similar to the motoneuron, BoNT is bound to receptors integrated into the liposome membrane. Via acidification of the outer milieu, the heavy chain translocates the light chain of the toxin into the liposome lumen. There, it cleaves a peptide reporter with a fluorophore and a quencher molecule attached to the opposite ends of its peptide chain. Upon cleavage of the peptide chain, the fluorophore is dequenched and yields a quantifiable fluorescent signal.
Figure 3
EM-characterisation of Fully Assembled Liposomes (FAL). Cryo SEM-picture of FAL taken in collaboration with Electron Microscopy ETH Zürich (EMEZ). Scale bar is 200 nm.
Method and Results
in progress (present status)
So far, reporter assays have been established for the sensitive detection of BoNT/A and BoNT/B, even under liposome-compatible conditions. Fully assembled liposomes (FAL) for detection of BoNT/A and BoNT/B have been successfully produced with lipid compositions similar to those found in mammalian nerve cells. FAL contain high amounts of the peptide reporter molecules and the respective receptors integrated into the liposomal membrane. First results indicate that the proposed detection system is functional (proof-of-concept). Currently, experiments are in progress to characterize the observed reaction kinetics and to optimize the system’s robustness and sensitivity.
Figure 2

Figure 3
Conclusions and Relevance for 3R
If the proof-of-principle is to be successful, then exact validation of the system’s capability will be the following step. If this demonstrates that the assay is sensitive and robust enough it may be used to replace the mouse LD50 test for batch control of pharmaceutical products that contain BoNT. Although the proof-of-concept aims to test for BoNT/B (active compound e.g. in Myobloc), modification of the assay may provide the means of detecting additional relevant BoNT types, especially BoNT/A (the active compound in BOTOX and others). Also, if the necessary receptors and substrates are available, it may theoretically also be used to detect Tetanus toxin, and even other toxins with multiple active sites which act in a way similar to BoNT.
References
1. EDQM (2011) 07/1022:2113 Botulinum toxin type A for injection. European Pharmacopoeia 72. pp. 3613-3615.
2. Schantz EJ, Kautter DA (1978) Microbiological methods: Standardized assay for Clostridium botulinum toxins. J AOAC 6: 96.
3. Bitz S (2010) The botulinum neurotoxin LD50 test - problems and solutions. ALTEX 27: 114-116.
4. Balls M (2003) Botulinum toxin testing in animals: the questions remain unanswered. ATLA 31: 611-615.
5. Adler S, Bicker G, Bigalke H, Bishop C, Blumel J, et al. (2010) The current scientific and legal status of alternative methods to the LD50 test for botulinum neurotoxin potency testing. The report and recommendations of a ZEBET Expert Meeting. ATLA 38: 315-330.
6. Chen C, Han D, Cai C, Tang X (2010) An overview of liposome lyophilization and its future potential. Journal of controlled release : official journal of the Controlled Release Society 142: 299-311.
7. Shulkin PM, Seltzer SE, Davis MA, Adams DF (1984) Lyophilized liposomes: a new method for long-term vesicular storage. J Microencapsulation 1: 73-80.
| Letzte Änderung: 12.10.2018 |