 |
3R-INFO-BULLETIN 55
October 2015
Author

Dr. Thomas Kaufmann is an associate professor at the Institute of Pharmacology, University of Bern. He is leading a team of 6-8 scientists with a research focus on the regulation of programmed cell death (PCD) and the contribution of de-regulated PCD to diseases such as cancer and immunological disorders. One cell type of interest is the basophil, which is a rare subtype of immune cell that is important in the defense against certain parasites, in immunoregulation, and in some forms of allergic reactions. To study the biology of these rare cells, the team of Prof. Kaufmann has developed a method to differentiate and massively expand murine basophils in vitro. For certain scientific questions, this approach permits a reduction in the number of experimental animals. Importantly, such basophil-committed cell lines can be generated from transgenic mice with embryonically lethal or severe phenotypes. Also, the amenability of the cells to further genetic manipulation, and the large number of cells that can be generated, render the model highly attractive for the study of molecular mechanisms (e.g., cell signaling pathways) in basophils.
Address:
Prof. Thomas Kaufmann
thomas.kaufmann@pki.unibe.ch
Institute of Pharmacology
University of Bern
CH-3010 Bern
Switzerland
Editor
Ernst B. Hunziker, Scientific Adviser of the 3R Research Foundation
Establishment of a novel system for the production of large numbers of mouse basophils in vitro
Basophilic granulocytes (basophils) are a rare leukocyte population within the blood, playing crucial roles in innate immune defense, immune regulation and allergic responses. The study of primary basophils is challenging owing to their low numbers and short life-span, as well as to the lack of suitable in-vitro cell-modeling systems and efficient differentiation protocols. In project No. 127-11 the research group of Thomas Kaufmann has developed a novel methodology whereby large numbers of mouse basophils can be differentiated in vitro. For specific scientific questions, this model could serve as an attractive alternative to primary cells in the study of basophil biology.
The study of primary basophils is a challenging endeavor
Basophils constitute less than 1% of the leukocyte population in peripheral blood. These specialized immune cells of the myeloid lineage play important roles in the defense against certain parasites, are important immune regulators, and are critically involved in some forms of allergic response (1). The study of primary basophils is a challenging endeavor in so far as these cells are end-differentiated (viz., do not multiply) and short-lived. Furthermore, their in-vitro differentiation from hematopoietic stem- or progenitor cells, which are typically isolated from bone marrow, is inefficient, and there exists no suitable cell-culture model for those of human or murine origin. Consequently, large numbers of mice have to be sacrificed to generate sufficient quantities of primary basophils for experimental purposes. With a view to sparing animal life, we have recently established a novel method to generate conditionally immortalized, basophil-committed progenitor cell lines. These progenitors can be induced in vitro to differentiate into mature basophils on demand and in well-nigh unlimited quantities (2).
Homeobox genes, such as Hoxb8, have the potential to block the differentiation of myeloid cells, which they often do in leukemias. We make use of this property to conditionally (reversibly) immortalize basophil-committed progenitors (Figure 1).
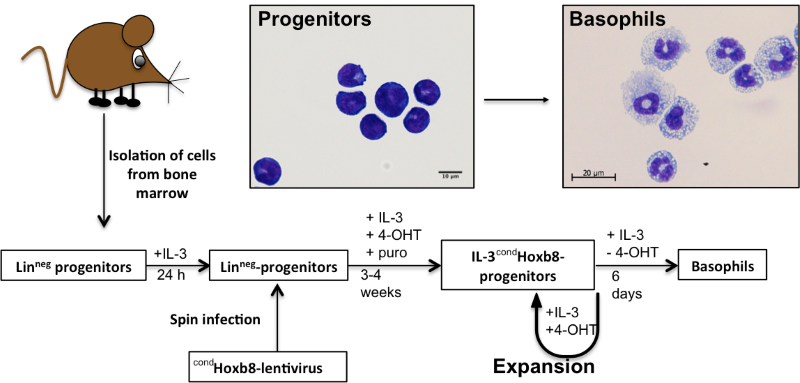
Fig 1.: Generation of conditionally immortalized, basophil-committed progenitor cell lines from the bone marrow of mice. IL-3: interleukin-3; Linneg: lineage surface marker negative; 4-OHT: 4-hydroxytamoxifen; puro: puromycin.
These basophil progenitor lines are generated from murine bone marrow (or fetal livers) using a significantly modified version of a protocol that has been published for the production of murine neutrophils/macrophages (2, 3). Established “Hoxb8 cell lines” can be maintained in culture for prolonged times and are amenable to further genetic manipulation using classical approaches - a feat that is very hard to achieve in primary granulocytes. Their differentiation into mature basophils is attained upon a shutdown of exogenous Hoxb8 expression. An enriched population of cells with the phenotypic and functional characteristics of mature basophils is obtained within 6 days. Mature Hoxb8 basophils are end-differentiated, have lobulated nuclei, a granular appearance, and express a number of cell-type-specific surface proteins (Fc□RIhighCD11b+ckitneg Gr1negIL5RnegCCR3neg).
Moreover, the basophil-specific mast cell proteases mMCP8 and mMCP11 are highly expressed in the differentiated basophils but not in their progenitors (Figure 2).
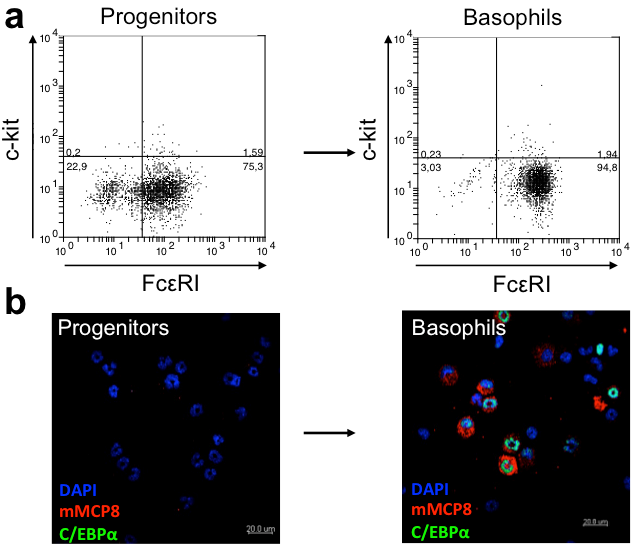
Fig 2.: a) Characterization of "Hoxb8 basophils" by flow cytometry. Basophils are characterized by their surface expression of the high-affinity IgE receptor, Fc□RI, and by the absence of staining for c-kit. b) Confocal microscopic images of differentiated Hoxb8 basophils (right) and their progenitors (left). The former but not the latter express high levels of the basophil-specific protease mMCP8 (red) and of the transcription factor C/EBP□ (green). Nuclear DNA is stained blue.
The high-affinity receptor for IgE - Fc□RI - is functional, since its crosslinkage leads to degranulation and the release of typical granular enzymes. IgE-dependent and –independent activation results in the release of allergic mediators, including histamine and leukotriene C4, and in the de-novo synthesis of large quantities of the Th2 cytokines interleukin-4 and IL-13 (2). The half-life of Hoxb8 basophils in culture is comparable to that of primary basophils and is likewise prolonged after adding interleukin-3 (a cytokine) to the medium (Figure 3).
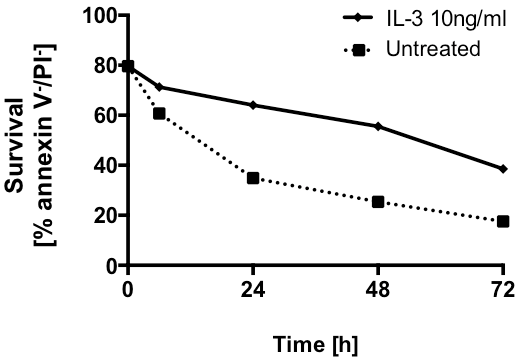
Fig 3.: Comparable to primary mouse basophils, the Hoxb8 variety is characterized by a relatively short half-life in culture, which was significantly prolonged after the addition of the cytokine interleukin-3 (IL-3). Cell viability (double negative annexin V/PI-population) was assessed by flow cytometry.
In summary, we have established a method to generate basophil-committed progenitor lines using Hoxb8. These progenitors can be induced in vitro to differentiate into mature basophils on a large scale, which, to the best of our knowledge, is a novel and unique attribute for a model of the kind. The here-described cell lines are genetically stable, can in principle be generated from any genetically modified strain of mice that survives up to embryonic stage E12, and can be easily handled in culture. Since large numbers of mice are required to isolate primary basophils, we would like to promote this cellular model as an alternative system to study specific aspects of basophil biology, and especially in instances when assays are employed for which large quantities of cells are called for. Importantly, Hoxb8 basophil lines can be generated from fetal livers (E12-E14), which would be a great asset when dealing with strains of mice displaying severe or even embryonically lethal phenotypes. Another considerable advantage of Hoxb8 progenitors over primary basophils is that they are amenable to efficient genetic manipulation using standard approaches, including the introduction of transgenes or the interruption of endogenous gene-expression according to either the CRISPR-Cas9 or the RNA-interference technology.
Relevance for 3R
The relevance of this project to the philosophy of the 3R Foundation therefore lies mainly in its potential to reduce not only the number of mice that are sacrificed for experimental purposes but also the need to breed mice with severe phenotypic disorders to the sentient stage of adulthood.
PDF version of this Bulletin No. 55
References:
- Karasuyama H, Mukai K, Tsujimura Y, Obata K. Newly discovered roles for basophils: a neglected minority gains new respect. Nat Rev Immunol 2009;9:9-13.
- Gurzeler U, Rabachini T, Dahinden CA, Salmanidis M, Brumatti G, Ekert PG, Echeverry N, et al. In vitro differentiation of near-unlimited numbers of functional mouse basophils using conditional Hoxb8. Allergy 2013;68:604-613.
- Wang GG, Calvo KR, Pasillas MP, Sykes DB, Hacker H, Kamps MP. Quantitative production of macrophages or neutrophils ex vivo using conditional Hoxb8. Nat Methods 2006;3:287-293.
| Dernières modifications: 11.10.2015 |