 |
3R-INFO-BULLETIN 19
January 2002
The Authors

Claudia Mertens (MSc) is an animal welfare expert of the SPCA in Zürich and member of two governmental 'Animal Care and Use Committees'. PD Dr. Rülicke is head of the Biological Central Laboratory and expert for transgenic technology at the Institute for Laboratory Animal Sciences University of Zürich, Switzerland. The project was carried out as a 'joint venture' by both authors.
Current address:
Claudia Mertens cmertens@smile.ch
Zürcher Tierschutz (Society for the Protection and Care of Animals)
CH-8044 Zürich
Switzerland
PD Dr. Thomas Rülicke truelick@bzl.unizh.ch
Biologisches Zentrallabor
Institut für Labortierkunde
Sternwartestr. 6
CH-8091 Zürich
Switzerland
Editor
Peter Maier, Scientific Adviser of the 3R Research Foundation
Phenotype Characterisation and Welfare Assessment of Transgenic Mice
Is there a particular health problem with transgenic animals?
Transgenic mice are valued models in biomedical research.[*] Several strains are used to investigate human congenital diseases; these strains carry a genetic defect that may or may not be clinically apparent in the animals. In practice, altering the genotype has no health consequences for about 90% of transgenic strains, but the remaining 10% have a reduced viability or impaired health at the phenotypic level (2). The impairment is also present even if the animals are not used for experimentation, and is transmitted from one generation to the next by normal breeding. The degree of impairment and the symptoms of suffering will vary (depending on the genetic alteration) from minor to severe and may show up at various stages of life in different strains. Within a single strain, the degree of suffering will depend primarily upon the genetic change and genetic background, but can also be influenced by housing and feeding conditions. It may or may not be possible to alleviate the suffering by specific measures. In any case, each newly created transgenic strain has the potential to cause poor health and suffering in the animals. In this regard, transgenic technology poses a challenge for the 3R goal of 'refinement': if a mouse strain is to carry certain dysfunctions at the genetic level for scientific reasons, everything has to be undertaken to minimise animal suffering.
Can transgenic animals offer a particular contribution to the goals of 3 R?
In spite of the problems listed above, transgenic animals may represent a refinement in comparison to some other traditional experimental models of disease in which the animals bear a heavy load of suffering. It seems possible to create 'elegant' models where - in the best case - a genotype is an excellent model of disease or dysfunction at the molecular or cellular level while the corresponding phenotype is completely healthy. However, in order to ensure that the well-being of a newly created transgenic strain is unaffected, a careful and comprehensive examination is necessary.
Given these two problems and tasks at the level of the animal strain - 1) recognition, prevention, minimisation and therapy of reduced welfare; 2) proof of absence of health problems due to a modified genotype - the first few generations of newly created mutant strains (founders, F1 and F2) need to be characterised with respect to their phenotype using health and welfare assessment guidelines.
What do we have to know about transgenic animals?
The aim of the project was to develop a standardised questionnaire for the characterisation of transgenic strains and a set of systematic score sheets to monitor the health of individual animals at different life-stages on a routine basis.
Forms for a standardised documentation of relevant information
The literature was first reviewed to collect clinical signs described as being useful for recognising health problems and suffering in laboratory animals. This body of literature was then analysed and a relatively large set of parameters selected for the characterisation of transgenic mice. The parameters had to fulfil two criteria: feasible for conducting as a screening test on a routine basis, and indicative of health or animal welfare problems at the strain level.
Next, two standardised forms were developed to characterise transgenic mice comprehensively (1). The form 'Data Record Form' (4, 6) includes 10 different score sheets for litter-wise and individual health monitoring from birth until spontaneous death or euthanasia. This information about individual animals was then structured and evaluated for the health and welfare status of the strain at two levels of detail in the form 'Strain Characterisation' (5, 6): Is there a welfare problem? (Basic Information). If so, give a detailed description of the symptoms (Detail Information; see Table 1 and 2).
Finally, the score sheets were tested for their "user-friendliness" and clarity in a pilot study that monitored and documented 106 mice from two established genetically modified strains in the first three months of life (4). The results demonstrated the fundamental practicality of the protocols, provided that personnel were adequately instructed and able to invest additional time in the monitoring programme.
Table 1 Form for strain characterisation: general structure
|
Table 2 Form for strain characterisation: basic information (C. Phenotype; overwiew)
|
Characterisation of transgenic animals improves animal welfare and science as well
A comprehensive characterisation of transgenic mice is indispensable for animal welfare, for the application of the 3R concepts, for the correct interpretation of research results, and for official purposes (annual statistics). The characterisation sheet presented here is feasible for routine use as a standardised procedure. It will contribute to increased scientific accuracy and efficiency in the laboratory. Transgenic animals, whether commercially distributed, transferred to new facilities or used in animal experimentation, should always be accompanied by their score sheet and strain characterisation form. Up to now, a lack of awareness of the problem and a lack of supporting regulations has prevented such a procedure from becoming standardised. However, this will change in the near future: the official characterisation form in use in Switzerland since 1998 (3) is currently under revision and the new version will be much more comprehensive.
References:
- The complete forms can be viewed as pdf.files ( "Charakterisierung", "Datenerfassungsformular") at http://www.bzl.unizh.ch/de/database/formtransg/index2.html (in German; the score sheets for individual monitoring - named "data record form" - are also available in English).
- Bundesamt für Veterinärwesen (2001): Statistik der Tierversuche in der Schweiz 2000. http://www.bvet.admin.ch/tierschutz/TV-Statistik/Jasta-00/D/Index.html
- Bundesamt für Veterinärwesen (1998): Gentechnisch veränderte Wirbeltiere: Anwendung der Tierschutzgesetzgebung auf Herstellung, Zucht, Haltung und Verwendung zu Versuchszwecken. in: Tierversuche und Alternativmethoden, Informationen und Richtlinien; Information Tierschutz 4.05. http://www.bvet.admin.ch/0_navigation-d/0_index-intern.html
- Mertens C. and T. Rülicke (1999): Score sheets for the monitoring of transgenic mice. Animal Welfare, 8; 433-438.
- Mertens C. and T. Rülicke (2000): Umfassendes Formular zur strukturierten Charakterisierung gentechnisch veränderter Tierlinien. ALTEX 17, 1/00; 15 - 21.
- Mertens C. and T. Rülicke (2000): Phenotype Characterization and Welfare Assessment of Transgenic Rodents (Mice). Journal of Applied Animal Welfare Science (JAAWS) 3(2); 127 - 139.
APENDIX:
Typical stages and corresponding handlings during individual welfare assessment and phenotype characterisation of mice:
1. First inspection of a litter within 24 hrs after birth
In addition to the usual control and registration of a newborn litter, maternal behaviour and viability of the neonates has to be closely observed. This includes 1) the assessment of the whole litter in the nest or cage (Fig. 1a) and 2) the assessment of each neonate on a hot plate (Fig. 1b). Peculiarities and abnormalities are recorded, litter mates are compared, body weight is measured from birth onward; a full stomach (Fig. 1c, the pup to the right) is an indicator for survival during the first few days. Dead pups are removed and stored for typing.

Fig. 1a
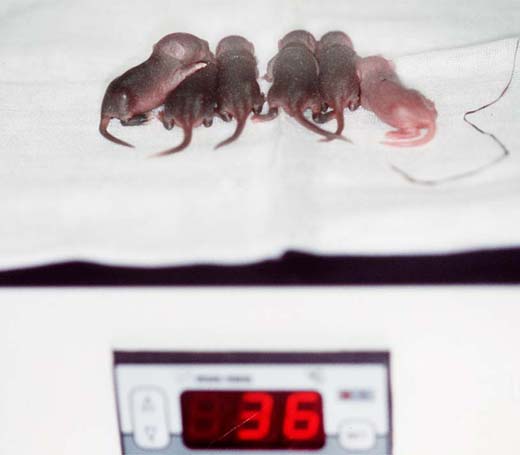
Fig. 1b
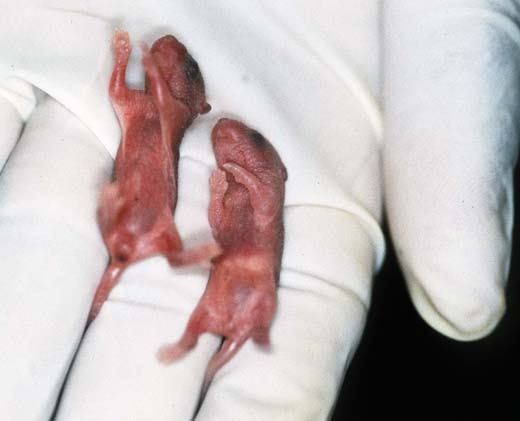
Fig. 1c
2. Ontogeny (from day 2 up to weaning)
Activity, reactivity and locomotion are important criteria to assess overall viability and vitality as well as the rate of development (Fig. 2a,b); these parameters can be observed without special testing. Neuro-behavioural tests like righting, forelimb grasping, pole grasping, vibrissa placing and auditorial startle are easy to perform with each pup (approximately day 10-14) and indicate rough senso-motor deficits. These tests are part of the standard screening for strain characterisation; Figures 2c,d show how the pole grasping test is performed, a test for motor (dis-)abilities and muscle strength. For further health monitoring the skin, the hair growth and all orifices have to be examined (2e,f). For ingestion after weaning the development of the teeth is important (Fig. 2f).

Fig. 2a
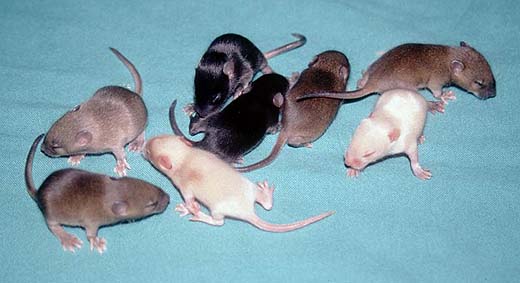
Fig. 2b
Fig. 2c
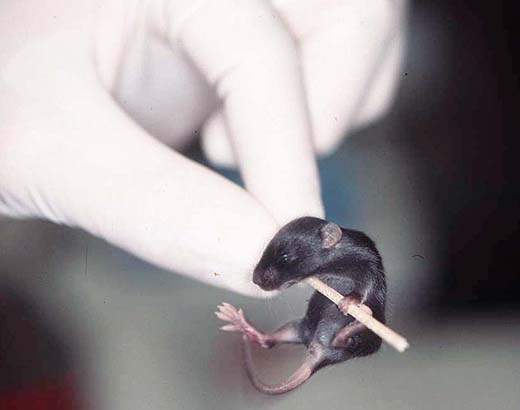
Fig. 2d
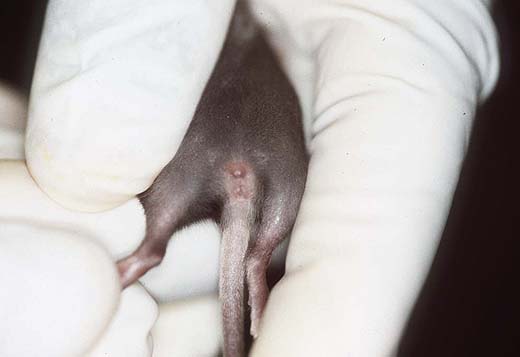
Fig. 2e
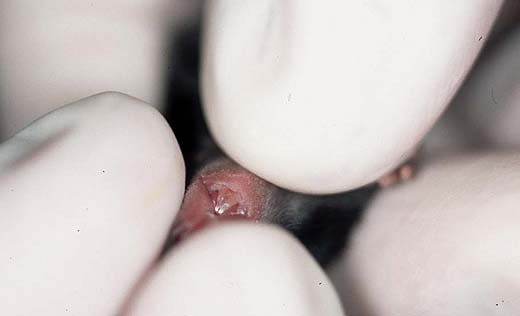
Fig. 2f
3. Technical procedures
A lasting individual identification of neonates by tatoo is necessary if the genotype of littermates used for strain characterisation might differ (3a). The usual identification methods for mice after weaning are ear punching or the implantation of a transponder (3b,c). At weaning (day 20) the sex of each pup is confirmed (Fig. 3d), females and males become separated and the sex ratio is determined (' reproduction data at population level). Technical procedures don't primarily serve welfare purposes; however, they are indispensable for phenotype characterisation and for individual monitoring from birth throughout the whole life.
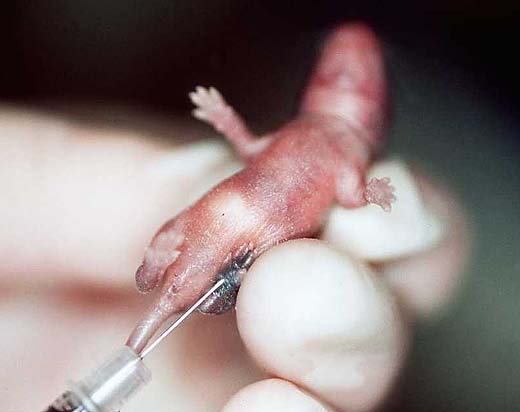
Fig. 3a
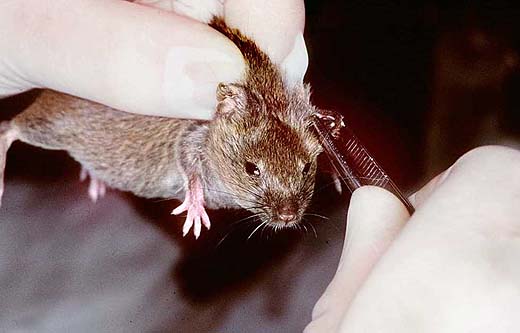
Fig. 3b
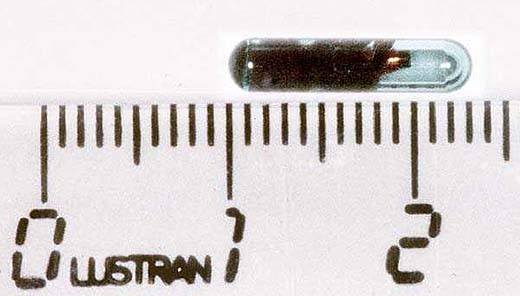
Fig. 3c

Fig. 3d
4. Virgin and adult animals (4th week up to spontaneous death or euthanasia)
The appearance of genetically modified animals can best be judged by comparison among littermates: (non-transgenic) wild type animals, heterozygous and homozygous transgenics (Fig. 4a; +/- and -/- littermates of the c-fos knock out are shown). All variations, e.g. differences in growth and development, are subject to further investigation of the strain. Observations have to be related to the exact genotype (hemizygous, heterozygous or homozygous) and if possible correlated with results of the expression analysis.
The observation of adult behaviour should include basic activities, inquisitiveness, posture and neuromuscular coordination. Note that obvious behavioral disorders such as locomotor stereotypes are more easily detected than the absence or inhibition of normal behaviour. Social behaviour like aggressiveness or nurture for offspring is not only indicative with respect to welfare, but also very important with respect to breeding strategies and the maintenance of a transgenic strain (Fig. 4b).
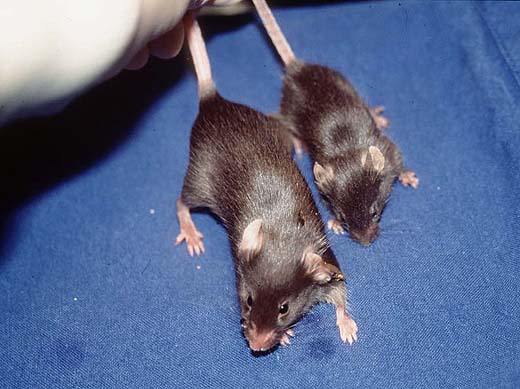
Fig. 4a

Fig. 4b
Published updated version of this Bulletin 19/2007 (PDF)
| [*] | Transgenic animals in animal experimentationVariations from the mean or norm have always been interesting to biomedical research in providing clues as to the normal and pathological functions of living beings. Genetic mutations are particularly useful because they can be reproduced by breeding; thus they are accessible to a thorough investigation. With the advent of transgenic technology and its possibilities of creating large numbers of mutant animals, the generation and use of genetically modified animals has increased dramatically in biomedical and pharmaceutical research and safety testing. In the year 2000, 59'000 such animals - mainly mice - were used for experimentation, an increase of 6.8% over the previous year. In the period 1997 to 2000, about 2600 transgenic mouse strains were being kept in Swiss laboratories. New strains are being added to this number continuously (2). |
| Dernières modifications: 30.01.2008 |