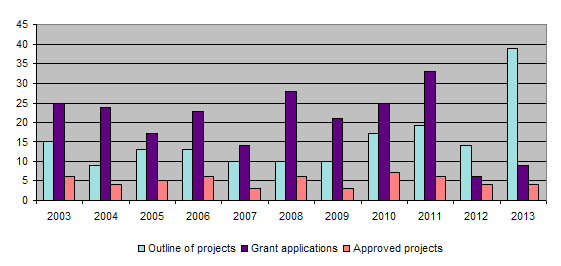

In 2013 the Foundation provided a total of Fr. 568,479.35 for 15 research projects. The Confederation and Interpharma made a total contribution of Fr. 730,000.00. The Administrative Board approved 4 new projects, while 3 projects were successfully completed. Out of the 39 project proposals submitted the Evaluation Committee selected 9 that were more relevant to the 3R principles and asked those applicants to submit a detailed description of their projects. A further selection process eliminated 5 projects whose relevance to the 3R principles was less evident, there being insufficient funds in the Foundation’s coffers for all nine. The 3R Info Bulletin 51 included the results of one project completed the previous year.
At the beginning of July 2013 the Scientific Advisor attended a conference oorganised by the Basle Declaration Society in London on the topic of "Transparency in animal research: Implementing openness in publication and communication". Discussion centred around ways to improve awareness of the 3R principles. Furthermore, Prof. Hunziker coordinated a working group of interested scientists whose aim was to draw up a proposal for a national 3R research programme to be submitted to the State Secretariat for Education, Research and Innovation.
Following the resignation of various members, five new people were elected to the Administrative Board during the year under review. As a result, from 2014 on, the fate of the Foundation will be in the hands of a Board consisting almost exclusively of new members under a new Chairman, namely Joachim Eder, a member of the Council of States.
In March 2013 Prof. Ernst Hunziker took over the role of Scientific Advisor and head of the Evaluation Committee. In December three new people joined the Committee, thus extending the scope of its expertise.
The 3Rs are Replace, Reduce and Refine animal experimentation. The 3Rs must be the guiding principles behind animal experimentation; if a study can be carried out without using any laboratory animals then such a procedure must be used. If it is essential to use laboratory animals under the terms of animal protection legislation the number used must be kept to a strict minimum. The third “R” requires that animals used for laboratory experiments be made to suffer an absolute minimum of pain and/or stress. The 3R Research Foundation funds research projects whose aim is to improve present-day experimental methods from the point of view of the 3Rs.
Detailed information about all the Foundation’s activities can be found on its website at www.forschung3r.ch.
A total amount of Fr. 568,479.35 was paid out for 15 ongoing projects during 2013.
Four new projects were approved in 2013 for which a total of Fr. 505,725 has been earmarked. These new projects are described in detail in the list of funded projects on the Foundation’s website (www.forschung3r.ch/en/projects/index.html).
In vitro engineering of a human cell-based three-dimensional dynamic model of atherosclerosis (135-13) Dr. Benedikt Weber, Swiss Centre for Regenerative Medicine, University Hospital Zürich, Switzerland. Cardiovascular diseases are a leading cause of death in the western world. In most cases, the underlying lethal pathology is atherosclerosis, which targets the walls of the blood vessels. In addition to a genetic predisposition, the disease is often associated with obesity, excessive stress and nicotine addiction. To promote the development of novel therapeutic drugs for the treatment of atherosclerosis, it is necessary to understand the factors that contribute to its onset and progression, for which purpose various animal models are employed. In this project, the aim is to elaborate a clinically relevant in vitro model of atherosclerosis using human cells that are derived from surgical waste material.
Development of an in-vitro potency assay for Clostridium chauvoei vaccines: Replacement of the guinea-pig-challenge potency (136-13) Prof. Joachim Frey, Institute of Veterinary Bacteriology, Vetsuisse Faculty, University of Berne, Switzerland. Blackleg is a bacterial disease that affects cattle and sheep. It leads to extensive tissue necrosis, particularly in the muscles of the limbs. For the development of new and/or improved vaccines to protect animals at risk, the efficacy of potential substances is tested in vivo. The process of screening is usually conducted on guinea pigs and is highly stressful for the animals involved. The aim of this project is to develop a safe in-vitro screening system for routine laboratory use that would render the ubiquitous guinea-pig-challenge potency test obsolete.
Validation of a new humanin-vitro model of microglia (137-13) Prof. Luis Filgueira, Department of Medicine, University of Fribourg, Switzerland. The local resident defence cells of the brain are microglia. They are implicated in inflammatory responses of the brain and in protecting it against infection, as well as in the regenerative processes that follow injury or pathological degeneration (such as that associated with Alzheimer’s disease). With a view to elaborating new therapeutic approaches to the treatment of brain diseases, the roles played by microglia in their development must be understood. Various animal models have been established to achieve this goal. Regrettably, even in-vitro studies cannot be conducted without the loss of animal life, since the necessary microglia can be obtained only from excised brains. The investigators who are working on this project have discovered that cells (monocytes) originating from human peripheral blood can be transformed into microglia. This finding opens up the possibility of establishing a human in-vitro model of microglia and thus of obviating the need for the sacrifice of animal life. In this project, the human in-vitro model of microglia will be validated.
Optimization of the nerve-cell-mimicking liposome assay as an in-vitro alternative for detecting Clostridium botulinum neurotoxins and validating their presence in complex sample materials (138-13) Marc-André Avondet and Prof. Stephen Leib, Toxinology Group, Spiez Laboratory, FOCP, Switzerland. Neurotoxins that are derived from the bacterium Clostridium botulinum are used medicinally to treat various disorders, such as dystonia, hyperhidrosis, strabismus, chronic pain and headaches. More recently, a lucrative market for these toxins has been captured in the field of cosmetic surgery, in which they are applied as anti-wrinkle agents. For the quality control of new toxin batches, more than half a million mice per year are utilized in Europe and the USA alone. With the ultimate goal of entirely replacing such mouse-based assays, the investigators working on this project will further develop a new methodological approach that has been conceived using nerve-cell-mimicking liposomes.
Generic in-vitro evaluation assay for immunological correlates of protection, to replace animal challenge infection (113-08) Prof. Artur Summerfield, Institute of Virology and Immunology (IVI), Mittelhäusern, Switzerland. The aim of this project was to provide a reliable and fast in-vitro test to replace in-vivo testing by which live animals suffer considerably through being exposed to vaccines in order to match a potential vaccine to the current serotype of foot-and-mouth disease virus in the field. The researchers were able to measure a response specific to the virus type in cultures of specialised immune cells. By using genetically modified cells it was possible to further simplify and improve the test. This provides a scientific basis for developing reliable tests based on cell lines. The new test will now be further validated by a European consortium (FMD-DISCONVAC).
Engineering of an in-vitro hepatocyte tissue system for malaria liver infection research (118-10) Dr. Dalu Mancama, CSIR, Biosciences Division, Pretoria, South Africa. With this project, the researchers aimed to develop a hepatocyte tissue culture system using human liver cells for the purpose of studying the primary infection process of malaria (infection with plasmodium sporozoites isolated from the Anopheles mosquito) and the proliferation of the parasites in cultured liver cells. The ultimate aim would be to develop vaccines, prophylactic medicines and an acute chemotherapy without using laboratory animals. The main stages of the study were successfully completed. The in vitro culture (proliferation) of the asexual stage of Plasmodium falciparum with erythrocytes, the culture of human hepatocytes with sporozoites and the co-culture with infected erythrocytes were all successful. The rate of infection observed was low and was influenced by the culture conditions. Further verification of the results will be carried out.
Comparative in-vitro and in-vivo testing of biofilm formation on the surface of bone grafts (124-10) Dr. Martin Clauss, Cantonal Hospital Liestal, Switzerland. The formation of bacterial biofilms on the inner and/or the outer bodily surfaces of humans and animals is a significant clinical problem, also in orthopaedics. Within such multilayered carpets, bacteria can establish a system of communication whereby the power of resistance to antibiotics is transmitted from one microbe to another. Moreover, the innermost parts of such biofilms are almost impenetrable to antibiotics, which greatly hampers the therapeutic fight against pathological bacteria. To date, research in this field has been conducted using animal models. With the ultimate goal of replacing experiments of this kind, the investigators engaged on this project wished to elaborate a novel in-vitro system for the simulation of bacterial films on bone-like surfaces. However, their endeavours met with no success.
3R-Info bulletins are published on the Foundation’s website (www.forschung3r.ch/en/publications/index.html).
Generic in-vitro evaluation assay for immunological correlates of protection in foot-and-mouth disease, to replace animal-challenge infections (no. 51, November 2013) Foot-and-mouth disease is an affection of viral origin, which attacks cattle and pigs. The consequences of infection are disastrous for individual animals. But also epidemiologically, an outbreak of the disease can spread like wildfire and is extremely difficult to eradicate. Hence, an effective vaccination of livestock is of paramount importance in controlling and combating outbreaks of the disease. The screening of potential vaccines is usually conducted in living animals and is a highly stressful experience for them. The aim of this project was to develop an in-vitro assay for the monitoring of vaccine potencies. Prof. Artur Summerfield’s research team at the Institute of Virology and Immunprophylaxis, Mittelhäusern, Switzerland have been successful in establishing a new in-vitro system to measure the efficacy of vaccines against foot-and-mouth disease, thereby obviating the need for conventional serological testing in animals.
The Foundation is a cooperative institution set up by the Parliamentary Group for Animal Experimentation Questions (public organ), Interpharma (association of pharmaceutical companies that carry out research, comprising at present AbbVie Ltd, Actelion Ltd., Amgen Switzerland Ltd, Bayer (Switzerland) Ltd, Biogen Idec Switzerland, Boehringer Ingelheim (Switzerland) Ltd, Bristol-Myers Squibb (Switzerland), Gilead Sciences Switzerland Ltd, Janssen-Cilag Ltd, Merck Serono International Ltd, Novartis Pharma Ltd, Pfizer Ltd, F. Hoffmann-La Roche Ltd, Sanofi-Aventis (Switzerland) Ltd, UCB Pharma Ltd and Vifor Ltd), and the Animalfree Research Foundation (animal protection). The Foundation was entered in the commercial register on 18 August, 1987.
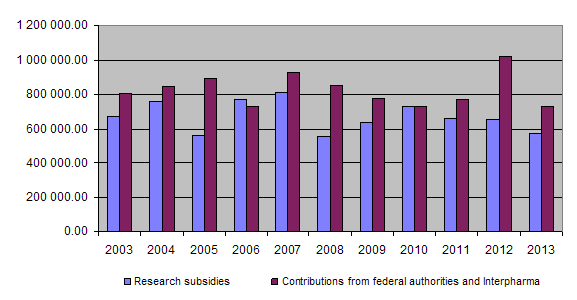
The funds for subsidising research are provided principally by the Federal Food Safety and Veterinary Office and Interpharma.
The purpose of the 3R Research Foundation Switzerland is to promote alternative research methods through grants for research projects as well as to implement and promote the 3R principles. The organisation supports first and foremost projects aimed at developing new methods or refining accepted methods (validation) which offer improvements vis-à-vis standard animal experimentation in line with the 3R motto, Replace, Reduce, Refine.
A broad range of projects is funded on the condition that they are likely to replace animal experimentation or to reduce the number of animals used or the stress and/or pain suffered. Accordingly, projects based on the Foundation’s three principles and covering any of a broad selection of bio-medical disciplines will be taken into consideration.
The Administrative Board of the Foundation is made up of nine members, two representing the Swiss parliament (1 seat vacant), two representing animal protection, two from Interpharma and two from the Federal Food Safety and Veterinary Office, as well as a representative of other interested circles. Current members are:
Christine Egerszegi, member of the Council of States, Mellingen (until 31.12.2013) (Chairwoman)
Joachim Eder, member of the Council of States, Unterägeri (Chairman as from 1.1 2014)
Dr. Peter Bossard, Horw (Deputy Chairman)
Dr. Philippe Bugnon, Institute of Laboratory Animal Science, University of Zurich (as from 1.11.2013)
Dr. Kaspar Jörger, Federal Food Safety and Veterinary Office, Berne-Liebefeld (as from 1.1.2014)
Dr. Ingrid Kohler, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Birgit Ledermann, Novartis Pharma Ltd, Basle (as from 1.6.2013)
Silvia Matile-Steiner, lawyer, Reinach (until 31.10.2013)
Claudia Mertens, biologist, Winterthur (as from 7.5.2013)
Dr. Markus Schmutz, Novartis Pharma Ltd, Basle (until 31.5.2013)
Nathalie Stieger, economist, F. Hoffmann-La Roche Ltd., Basle
Prof. Hans Wyss, Director of the Federal Food Safety and Veterinary Office, Berne-Liebefeld (until 31.12.2013)
Prof. Ernst B. Hunziker, University of Berne (as from 1.3.2013) (Chairman)
Prof. Hans Acha-Orbea, Department of Biochemistry, University of Lausanne (as from 1.11.2013)
Dr. Franziska Boess, F. Hoffmann-La Roche Ltd, Basle
Prof. Clemens A. Dahinden, Institute of Immunology and Allergology, University Hospital, Berne (until 29.5.2013)
Dr. Urban Deutsch, Theodor-Kocher-Institute, University of Berne (as from 1.11.2013)
Prof. Marianne Geiser Kamber, Institute of Anatomy, University of Berne
Prof. Andrew Hemphill, Institute of Parasitology, University of Berne
Prof. Simon P. Hoerstrup, Swiss Centre for Regenerative Medicine (SCRM),
University Hospital Zurich
Dr. Ingrid Kohler, Federal Food Safety and Veterinary Office, Berne-Liebefeld
Dr. Kurt Lingenhöhl, Novartis Pharma Ltd, Basle
Prof. Thomas Lutz, Institute of Veterinary Physiology, University of Zurich
Prof. Alex Odermatt, Department of Pharmaceutical Sciences, University of Basle (as from 1.11.2013)
Dr. Martin Reist, Sanisys Ltd, Biel (until 30.4.2013)
Dr. Stefanie Schindler, Animalfree Research Foundation, Berne
Prof. Ernst B. Hunziker, University of Berne (as from 1.3.2013)
Ernst P. Diener, lawyer, Münsingen
Waber Treuhand GmbH, Einigen
Federal Department of Home Affairs
In May 2013 Claudia Mertens, a biologist who works with the Zurich Animal Protection League, was elected to the Administrative Board to replace Dr. Franz Gruber, who resigned at the end of 2012 for reasons of age. June 2013 saw the election to the Board of Dr. Birgit Ledermann, Animal Welfare Officer at Novartis Pharma in Basle, as the representative of the pharmaceutical industry. She replaces Dr. Markus Schmutz, who also retired for reasons of age and received the Board's best wishes. At the end of October, the Foundation was pleased to welcome Dr. Philippe Bugnon, an expert with a proven track record in laboratory animal science, to the Administrative Board to represent other areas. He replaces Silvia Matile-Steiner, who took retirement. She had been on the board since 2008 and was warmly thanked for her hard work in relation to the Foundation's strategic concerns. Joachim Eder, a member of the Council of States, joined the Administrative Board as its new Chairman on 1 January 2014, accompanied by Dr. Kaspar Jörger, representing the Federal Food Safety and Veterinary Office. Their predecessors, Christine Egerszegi, who had been on the Administrative Board since 1999 and held the position of Chairwoman since 2008, and Prof. Hans Wyss, who joined the board in 2003 as the Director of the Federal Veterinary Office, were warmly thanked for their hard work and commitment by the Deputy Chairman.
In March 2013, Prof. Ernst Hunziker from the University of Berne took over as the Scientific Advisor and Chairman of the Evaluation Committee. The year under review saw the resignation of Prof. Clemens Dahinden from the Institute of Immunology and Allergology, University Hospital of Berne and Dr. Martin Reist from the Evaluation Committee; both were thanked for their contribution to the work of the Foundation. Three new faces joined the Evaluation Committee over the year, namely Prof. Hans Acha-Orbea from the Institute of Biochemistry at the University of Lausanne, Dr. Urban Deutsch from the Theador Kocher Institute of the University of Berne, and Prof. Alex Odermatt of the Department of Pharmaceutical Sciences, University of Basle.
In the Foundation’s twenty-seventh year of existence the Administrative Board met three times, namely in May, October and December, for a half-day meeting. Apart from the statutory business concerning the end of the business year 2012, the Board addressed the following issues.
In May, the Board focused on the financial statements for 2012 and the earmarking of research funds for ongoing projects. In addition, it took note of the final reports on three completed projects. Claudia Mertens and Birgit Ledermann were elected to fill the two vacancies on the Board.
At its meeting in October, the focus was on funding for new projects. Out of a total of 9 project proposals selected by the Evaluation Committe from the 39 applications received, 4 were finally approved. As far as concerns funding for the Foundation's activities, the Administrative Board were delighted to learn that Interpharma has renewed its promise of future financial support, which will ensure the further work of the Foundation in the coming years.
At the Administrative Board’s December meeting, which was also attended by members of the Evaluation Committee, the focus was on the outgoing Chairwoman Christine Egerszegi and Prof. Hans Wyss, who also left the Administrative Board at the end of the year, as well as the election of Joachim Eder as the Foundation's new Chairman. The meeting finished with a review of activities in 2013, followed by a convivial dinner.
With its input over the last few years concerning questions about the Foundation's activities, the Strategy Committee set up by the Administrative Board now considers its role fulfilled. At its May meeting, the Administrative Board warmly thanked the member of the Committee for all their hard work and officially declared the Committee disbanded.
During the year, the Administrator provided support for the new Scientific Advisor in taking over his tasks. His role is to deal with all matters concerning the Foundation that cannot be passed on to anyone else. In particular, he prepares all the necessary information for the Administrative Board to take their decisions, as well as dealing with correspondence with applicants and project managers. He also deals with payments, book-keeping, closing the books at the end of the financial year and the budget. In addition, he prepares the Annual Report as well as texts for the Foundation’s website.
Under the chairmanship of the Scientific Advisor, the Evaluation Committee held two meetings during the year, where in particular they examined 39 new applications for funding of which they chose 9 likely projects; finally 4 were approved. In addition they examined the final reports on 3 completed projects and submitted them to the Administrative Board. We should like to take this opportunity to thank the members of the Evaluation Committee for their voluntary work in this connection.
The new Scientific Advisorduly took over his tasks, which include preparing the meetings of the Evaluation Committee, publishing the 3R Info Bulletins (on the Foundation’s website at www.forschung3r.ch), writing the brief scientific reports in English which present the projects receiving funding on the Foundation’s website and regularly updating these reports. He was also kept busy assessing project proposals, reading intermediate reports, advising applicants and project managers, dealing with enquiries and explaining why projects had been rejected, as well as coordinating a working group set up to prepare a proposal for a national 3R research programme. Finally, he represented the Foundation at a conference organised by the Basle Declaration Society in London on the topic of: Transparency in animal research: Implementing openness in publication and communication.

During 2013 three projects were completed (113-08, 118-10, 124-10). Together with those projects completed earlier, this brings the total of finished projects to 120 out of 138.
The bar-chart shows that the number of project proposals increased considerably in 2013. In 2012, the call for projects was restricted to one deadline only, namely in February 2012, and a new two-stage application procedure was introduced. Initially, applicants submit a project proposal, 39 such proposals being received in 2013. After the proposals have been examined by the Evaluation Committee, the latter then asks those whose proposed projects are relevant to the 3R principles to draw up detailed applications, 9 such detailed descriptions being submitted in 2013. Out of these 9 proposals, the Administrative Board finally approved 4 applications. Regardless of the many project proposals received, there is little fluctuation in the number of projects approved for funding owing to the limited means available to the Foundation.
A total of Fr. 572,162.35 was paid out for research in 2013. Operational expenditure for the year amounted to Fr. 196,028.10 (project monitoring and information Fr. 89,014,10, administrative costs including office infrastructure Fr. 107,014,00). Total expenditure therefore amounted to Fr. 768,190,45.
On the income side, the equal financial commitment of the federal authorities and Interpharma represent the basic funding for the Foundation’s activities, each providing in 2013 an amount of Fr. 365,000. Further income of Fr. 1,680.85 was accrued.
Total income was therefore Fr. 731,680.85 while total expenditure amounted to Fr. 768,190.45, giving an excess of expenditure over income of Fr. 36,509.60. The unused funds item fell from Fr. 287,870.96 at the end of 2012 to Fr. 251,361.36 at the end of 2013, the latter figure constituting the Foundation’s new reserve of liquid assets.
At the end of 2013 the total earmarked for projects approved by the Board but not yet paid out amounted to Fr. 835,239.35. This future liability is covered by Interpharma’s promise of funding VI together with the contribution from the Confederation. The Foundation’s credit with Interpharma amounted to Fr. 2,747,000 at the end of 2013.
The budget for 2014 includes Fr. 409,046.35 for current projects and a maximum amount of Fr. 600,000 for new projects.
At the end of 2013 a total of Fr. 18,852,941 had been granted for projects and other subsidies, of which Fr. 18,017,701.65 had been paid out so far. Together the federal authorities and Interpharma have contributed Fr. 21,696,000 to the Foundation since 1987.
| Profit and loss account 2013 | Expenditure | Income | |
|---|---|---|---|
| Income | |||
| Federal contribution | 365 000.00 | ||
| Contribution from Interpharma | 365 000.00 | ||
| Total contributions | 730 000.00 | ||
| Capital yield | 375.15 | ||
| Reimbursement of research grants | 0.00 | ||
| Other income | 1305.70 | ||
| Total income | 731 680.85 | ||
Expenditure | |||
| Research grants | 572 162.35 | ||
| Project supervision and information | 89 014.10 | ||
| Administrative expenses | 107 014.00 | ||
| Total expenditure | 768 190.45 | ||
| Excess expenditure over income | - 36 509.60 | ||
| 731 680.85 | |||
| Balance as per 31st December 2013 | Assets | Liabilities | |
| Liquid Assets | |||
| Bank | 259 826.78 | ||
| Accounts payable | 274.63 | ||
| Accounting apportionment assets | 1 542.75 | ||
Liabilities | |||
| Accounting apportionment liabilities | 9 282.80 | ||
| Unused research funds | |||
| Carried forward 1. 1. 2013 | 287 870.96 | ||
| expend. over income | -36 509.60 | 251 361.36 | |
| Capital | 1 000.00 | ||
| 261 644.16 | 261 644.16 | ||
Contingent liabilities | |||
| Approved research grants not yet paid out | Sfr. 835 239.35. | ||
Münsingen, 15 March 2014
3R RESEARCH FOUNDATION
Chairman: signed Joachim Eder
Administrator: signed Ernst P. Diener
Waber Treuhand GmbH in Einigen audited the financial statements for the year according to standards of limited auditing and did not find any indication that the accounts and statements do not correspond to current legislation or the principles and regulations of the Foundation.