 |
3R-Project 133-12
Development of an in vitro system to grow and investigate vascular endothelial cells under physiological flow conditions
Riccardo Sfriso, Shengye Zhang, Oliver Steck, Robert Rieben1
1Department for BioMedical Research, University of Bern, Switzerland
robert.rieben@dkf.unibe.ch
Keywords: endothelia; coagulation; transplantation; ischemia; reduction; replacement; drug screening
Duration: 2 years Project Completion: 2016
Background and Aim
Activation of the vascular endothelium is an important mechanism underlying the pathophysiology of several clinical conditions such as ischemia/reperfusion injury (IRI), organ rejection as well as cardiovascular diseases in general. The maintenance of an anti-inflammatory, anti-thrombotic and pro-fibrinolytic quiescent endothelium is the key to prevent the onset of such conditions. However, the molecular mechanisms leading to endothelial cell activation are difficult to investigate in vivo and in order to reduce the number of animal experimentations in vitro assays are highly desirable.
Nowadays endothelial cells from different vessels are easily isolable and cultivable. However, current standard in vitro assays are often limited in terms of structural complexity, surface to volume ratio and shear stress, which are typical features of the in vivo situation.
Based on the 3R principles we have developed a novel 3D microfluidic in vitro model which aims to reproduce the physiological situation present in small to medium sized vessels.1-3 Endothelial cells cultured on the lumen of artificial round-section microvessels are exposed to flow and physiological shear stress, allowing a better reproduction of what endothelial cells are facing in vivo. Such a model permits to reach an adequate endothelial cell surface to volume ratio for the study of the natural anti-coagulant and anti-inflammatory properties of the endothelium. The system can be used to test the effects of different drugs on the endothelium as well as to screen the effects of human transgene expression in porcine endothelial cells for xenotransplantation.
Method and Results
in progress (present status)
The 3D in-vitro model allows to build a monolayer of endothelial cells on the inner layer of round section microchannels (Fig.1), which closely mimic the structure and geometry of in vivo vessels. On the microchannels endothelial cells are exposed to pulsatile flow and physiological shear stress.
In a xenotransplantation setting the monolayer of endothelial cells will be exposed to human serum, plasma or whole blood. Immunoglobulin binding, complement deposition, coagulation markers as well as endothelial cell activation can be assessed by immunofluorescence and high resolution confocal microscopy. Soluble cytokines and complement activation markers can be assessed in the fluid phase.2,3
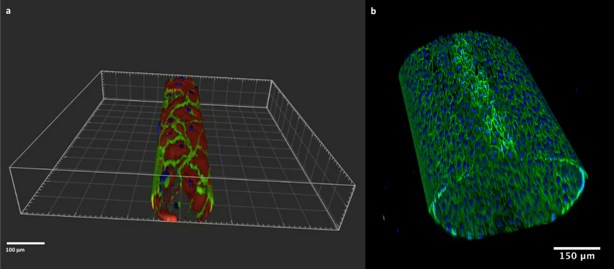
Figure 1: Artificial microchannels of 100 µm (left) and 550 µm (right). VE-cadherin (green) and F-Actin (red) are stained. Nuclei are stained with DAPI (blue).
Conclusions and Relevance for 3R
Currently, the model has been validated and well characterized. The data obtained after perfusion with human serum are available and current preliminary data after perfusion with whole blood are convincing. In the context of xenotransplantation, the model is helpful for the testing of multiple human transgenes prior to the production of pigs for in vivo xenotransplantation. The model will thus help to reduce the number of animals for in vivo experimentation.
References
1. Sfriso, R., Zhang, S., Bichsel, C. A., Guenat, O. T. & Rieben, R. A vessel-like microfluidic system to study complement and coagulation in the context of xenotransplantation. Immunobiology 221, 1175 (2016).
2. Sfriso, R. et al. In vitro assessment of xenogeneic complement and endothelial cell activation using a microvascular flow model. Mol. Immunol.89, 209 (2017).
3. IXA 2017- Abstracts of the 14th Congress of the International Xenotransplantation Association, Baltimore, USA. Xenotransplantation 24, e12328–108 (2017).
| Dernières modifications: 12.10.2018 |