 |
3R-Project 129-11
The use of microfluidic chambers to study axonal transport in PTEN and SOCS3 dependent axonal regeneration
Zhigang He and Thomas L. Schwarz
F.M. Kirby Neurobiology Center and Children's Hospital and Department of Neurology, Harvard Medical School
Boston, MA 02115, USA
zhigang.he@childrens.harvard.edu, thomas.schwarz@childrens.harvard.edu, romain.cartoni@childrens.harvard.edu
Keywords: mice; axons; neurons; spinal cord; spinal cord repair / diseases; perfusion chamber; reduction; replacement
Duration: 1 year Project Completion: 2013
Background and Aim
Rodents are extensively used to study nerve injury. The mouse spinal cord injury model, widely used in nerve injury research, is extremely debilitating. In vivo studies have allowed major advancements in the comprehension of the incapacity of adult central nervous system axons to regenerate. Notably, in vivo studies have shown that axonal regeneration after nerve injury was possible in adult mice if PTEN or SOCS3 were deleted in knock-out mice (1,2,3). However, studying in vivo mechanistic adaptation at the cellular level remains challenging. Axonal transport is an important cellular mechanisms evidenced by the numerous neurodegenerative diseases that have been related to an axonal transport impairment. Improving axonal transport in the injured and diseased central nervous system has been proposed as a promising strategy to improve neuronal repair. However, the contribution of each cargo to the repair mechanism is unknown. Because the transport of specific cargos after axonal insult has not been examined systematically in a model of enhanced regenerative capacity, it is unknown whether the transport of all cargos would be modulated equally in injured central nervous system neurons. In order to test the transport adaptation of cellular cargos during axonal regeneration, an in vitro system allowing straightforward manipulation and analysis is required. We used microfluidic chambers (4) to mimic nerve injury and regeneration in vitro. This method allowed us to (i) injure axons without affecting the cell body, (ii) manipulate neuron cell bodies and axons specifically, and (iii) analyze axonal transport during axonal regeneration at a single axon resolution.
Method and Results
Using a microfluidic culture system we compared neurons co-deleted for PTEN and SOCS3, an established model of high axonal regeneration capacity, to control neurons. We measured the axonal transport of three cargos (mitochondria, synaptic vesicles and late endosomes) in regenerating axons and found that the transport of mitochondria, but not the other cargos, was increased in PTEN/SOCS3 co-deleted axons relative to controls. The results reported here suggest a pivotal role for this organelle during axonal regeneration and validate the microfluidic culture system to identify cellular mechanisms occurring in regenerating axons.
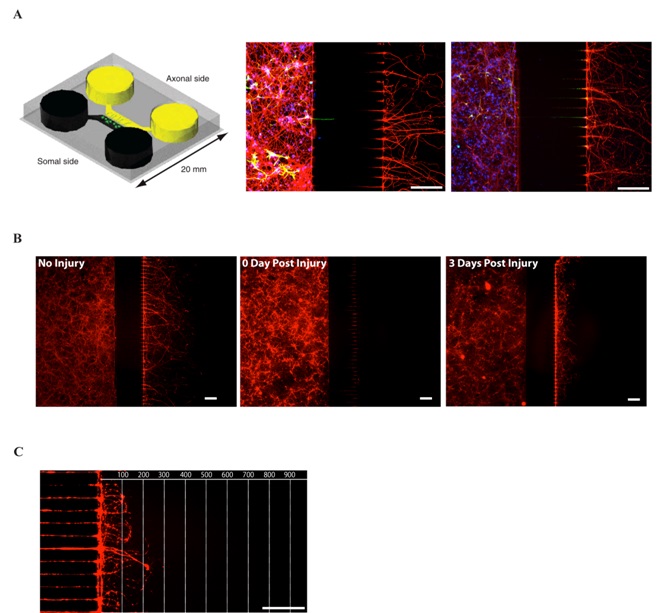
Figure 1: The microfluidic culture system
(A) First panel: Schematic view of a microfluidic chamber. Adapted from (4). Neurons plated in the somal side will project their axons through the 450 µm long microgrooves and reach the axonal side. Second and third panel: immunohistochemistry of E18 mouse cortical neurons culture (DIV7) in microfluidic chambers. Second panel: Red: anti-3-Tubulin (axonal marker), Green: anti-GFAP (glial marker), Blue: DAPI (nuclear marker). Third panel: Red: anti-3-Tubulin, Green: anti-MAP2 (dendrites marker), Blue: DAPI. 450 µm microgrooves allow a pure isolation of neurons.
(B) Tuj1 immunohistochemistry of E18 mouse cortical neurons culture (DIV7) in microfluidic chambers. No Injury (first panel), immediately after injury (second panel) and 3 days after injury (third panel).
(C) Regrowing axons can be observed 3 days after injury.
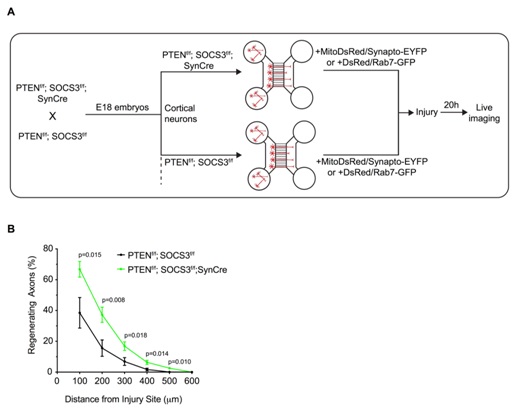
Figure 2: Deletion of PTEN and SOCS3 improved axonal regeneration of E18 cortical neurons
(A) Schematic of the in vitro platform to study the axonal transport in regenerating PTEN-/-; SOCS3-/- cortical neurons. PTEN-/- ; SOCS3-/- E18 cortical neurons were obtained by breeding PTENf/f ; SOCS3f/f mice with the PTENf/f; SOCS3f/f; Synapsin Cre (SynCre) mice. The cortex of each embryo was processed individually so that each microfluidic chamber was seeded with neurons from a single embryo. Thereby PTENf/f; SOCS3f/f; SynCre neurons were compared to PTENf/f; SOCS3f/f from littermate embryos. All neurons were cotransfected with MitoDsred2 and EYFP-Synaptophysin or Rab7-GFP.
(B) Quantification of in vitro axonal regeneration of PTEN-/- ; SOCS3-/-and controlcortical neurons 20h post injury. n=9-11 microfluidic cultures of individual embryos from 5 independent experiments. Two tailed Student’s Unpaired t-test.
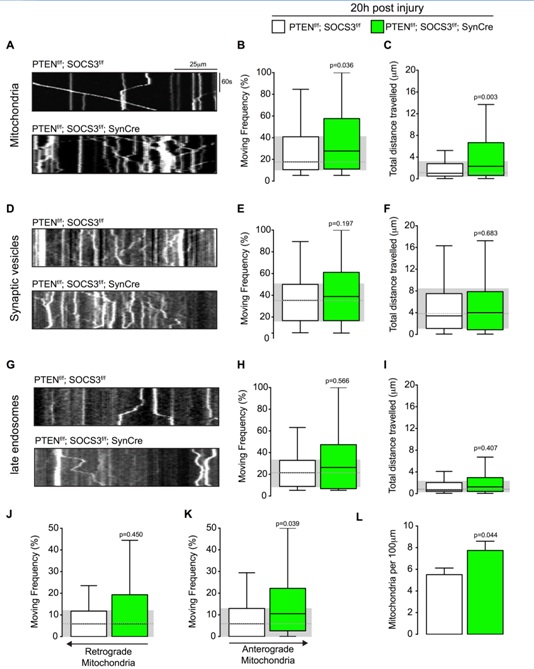
Figure 3: Mitochondrial transport is increased in regenerating axons co-deleted for PTEN and SOCS3.
(A) Representative kymographs from live imaging of mitochondria in regenerating axons from PTENf/f ; SOCS3f/f and PTENf/f ; SOCS3f/f; SynCre neurons 20 h post injury. Consecutive line scans of the axon were arrayed top to bottom so that the y-axis of the kymograph represents the time and the x-axis the position of the object studied. Stationary objects therefore appear as vertical lines and motile ones as diagonals.
(B, C) Box plot showing the moving frequency of motile mitochondria (B) and their distance travelled (C) in regenerating axons of the indicated genotypes. Mann-Whitney U test on the number of mitochondria. n=136-223 mitochondria from 18-22 axons and 4-6 individually cultured embryos from 2 independent litters. (D-F) Representative kymographs (D) and quantification of the moving frequency (E) and distance travelled (F) from live imaging of synaptophysin-positive synaptic vesicle precursors in regenerating axons of the indicated genotypes. Mann-Whitney U test. n=153-226 synaptic vesicles, 12-17 axons, 4-5 individually cultured embryos from 2 independent experiments. (G-I) Representative kymographs (G) and box plots of moving frequency (H) and total distance travelled (I) from live imaging of late endosomes in regenerating axons of indicated genotype. (Mann-Whitney U test. n=69-105 late endosomes, 13-14 axons, 5-4 individually cultured embryos from 2 independent experiments. (J and K) Retrograde (J) (toward cell body) and anterograde (K) (toward axon’s tip) moving frequencies of the mitochondria analyzed in (A). Mann-Whitney U test. (L) Mitochondrial densities in the axons analyzed in (A). Two tailed Student’s Unpaired t-test. Data in all the box plots are represented with a box that delimitates the lower (Q1) and the upper quartile (Q3) of the distribution. Horizontal line indicates the median (Q2) and whiskers indicate the maximum and minimum of the distribution.
Conclusions and Relevance for 3R
A lab testing the regenerative capacity of axons using the spinal cord injury model (transgenic mice, drug treatment) will use roughly 5,000 mice per year. Some of these mice are used to test hypotheses that will not give any satisfactory results. To increase the chance of obtaining positive results in vivo while decreasing the number of mice used, we propose to validate the microfluidic chambers as an in vitro system that would be a primary test to establish promising hypotheses worth testing in vivo, if possible. We estimate that by first testing the hypotheses in a reliable in vitro system would save one third of the mice used per year. We hope that our study will establish microfluiding chambers as a gold standard in vitro system in the field of the study of spinal cord injury/axonal regeneration.
The results of this project is published in:
Cartoni, R., Pekkunaz, G., Wang, C., Schwarz,T.L., He, Z. An Elevated Mitochondrial Transport Rate characterizes high Regeneration Capacity Neurons in CNS neurons (2017). PLoS One 12(9).
References
(1) Park K, Liu K, Hu Y, Smith P, Wang C, Cai B, et al. Promoting axon regeneration in the adult CNS by modulation of the PTEN/mTOR pathway. Science. Vol 322, 2008b: 963-6.
(2) Smith P, Sun F, Park K, Cai B, Wang C, Kuwako K, et al. SOCS3 deletion promotes optic nerve regeneration in vivo. Neuron. Vol 64, 2009: 617-23.
(3) Sun F, Park KK, Belin S, Wang D, Lu T, Chen G, et al. Sustained axon regeneration induced by co-deletion of PTEN and SOCS3. Nature; 480: 372-5.
(4) Taylor AM, Blurton-Jones M, Rhee SW, Cribbs DH, Cotman CW, Jeon NL. A microfluidic culture platform for CNS axonal injury, regeneration and transport. Nat Methods. Vol 2, 2005a: 599-605.
| Dernières modifications: 12.10.2018 |