
The Foundation is a cooperative institution set up by the Parliamentary Group for Animal Experimentation Questions (public organ), Interpharma (Novartis Pharma Ltd, F. Hoffmann-La Roche Ltd, Serono Ltd) and the Foundation for Animal-Free Research (animal protection). It was entered in the commercial register on 18th August, 1987.
The funds provided to subsidise research stem from the Federal Veterinary Office and Interpharma.
The purpose of the 3R Research Foundation Switzerland is to promote alternative research methods which avoid the use of animals through grants for research projects. The organisation supports first and foremost projects aimed at developing new methods or refining accepted methods (validation) which offer practical improvements vis-à-vis standard animal experimentation in line with the 3R motto Reduce, Refine, Replace.
A broad range of projects is sponsored on the condition that they are likely to reduce the number of animals used or the stress and/or pain suffered. Projects considered must be based on the Foundation's three principles and are mainly in the bio-medical multidisciplinary field.
In its fifteenth year of existence the Administrative Board met twice, in April and December, for a half-day meeting. Apart from the statutory business concerning the end of the business year 2000, the Board addressed the following issues.
Research funds for 2001 were allotted to 12 projects already underway. In addition, 5 new projects were approved, while 12 applications were rejected. The Board also took note of the final assessment by the Evaluation Committee of 6 projects which had been completed. In April the necessary funds were approved for setting up an Internet solution for 3R training.
In order to pursue the Foundation's activities, the Board decided in December to submit a new application for funds to Interpharma.
In October 2001 the 3R Research Foundation became a member of the European Consensus Platform for Alternatives (ECOPA) in Brussels. This means that the Foundation is now part of a European network, to which only EU member states normally have access.
With the support of the scientific advisor, the Evaluation Committee held two meetings during the year, where they assessed new applications and evaluated completed projects. The Scientific Advisor's main task was to publish the 3R Info Bulletin (as a brochure and on the Foundation's Internet site at www.forschung3r.ch), as well as writing brief scientific reports in English which present funded projects on the Foundation's Internet site. Much time was also devoted to advising applicants and project leaders, calling for intermediate reports, evaluating draft projects and dealing with enquiries and explanations for the rejection of applications. He also played a major role in designing and realising the planned Internet training module for 3R requirements (further training for specialists who carry out experiments involving live animals). Finally, he represented the Foundation at several meetings in Switzerland and abroad. As part of a European working group, he was involved in the preparatory work for setting up ECOPA.
During the year 4 projects were completed: 54/96, 65/98, 68/99 and 74/00. Together with those projects completed earlier (1-5/87, 6-15/88, 16/89, 17-20/90, 21-24/91, 25-42/92, 43-44/95, 45-51/96, 53/96, 55/96, 57/97, 59-62/97 64/97) this brings the total of finished projects to 63 out of 80.
In 2001 there were no changes in the Board nor the Evaluation Committee
Prof. Peter Maier was taken on part-time as Scientific Adviser.
So far the following 3R Information Bulletins have been published in English:
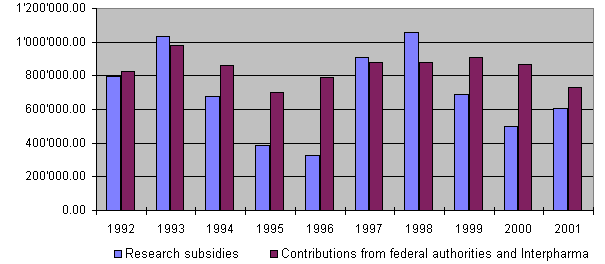
During 2001 around Sfr. 586,000 was used for research grants. An additional Sfr. 15,000 repre-sented an initial payment for setting up an Internet solution for 3R training, while around Sfr. 4,500 was used for setting up ECOPA. Project supervision and information accounted for around Sfr. 91,000 and administrative expenses totalled Sfr. 73,000. Total expenditure was therefore around Sfr. 770,000. The amount spent on research for current projects (Sfr. 586,000) was thus approximately Sfr. 68,000 below budget. This is mainly due to deferred payments in connection with final reports, the 5% reserve not yet having been allotted or paid out. In addition, a budgeted amount of Sfr. 100,000 was not used because two projects are due to be launched only in 2002; on the other hand, an amount of Sfr. 159,000 has already been transferred for three new projects. Operational expenditure for project supervision, information and administration totalled around Sfr. 165,000 and was thus Sfr. 3,000 over budget.
On the income side, the equal financial commitment of the federal authorities and Interpharma constitutes the basis for the Foundation's activities. Each body donated a total of Sfr. 365,000 to 3R. The additional payment from the Confederation of Sfr. 80,000, transferred at the end of the year for 2002 is included in the liabilities accounting apportionment and will be credited in the financial statements for 2002.
Income totalled around Sfr. 739,000 (federal authorities and Interpharma together Sfr. 730,000, interest on bank account Sfr. 9,000) while total expenditure amounted to Sfr. 770,000, which gives a negative balance of approximately Sfr. 31,000. The balance of unused research funds therefore fell from around Sfr. 520,000 at the end of 2000 to Sfr. 489,000 at the end of 2001.
The budget for the year 2002 includes a sum of around Sfr. 777,000 for current projects and a maximum of Sfr. 360,000 for new projects.
By the end of 2001 a total of Sfr. 11,198,421.86 had been granted for projects and other subsidies, of which a total of Sfr. 10,056,404.81 has been paid out so far. Together the federal authori-ties and Interpharma have contributed Sfr. 11,726,000 to the Foundation since 1987.
| Profit and loss account 2001 | Expenditure | Income | |
|---|---|---|---|
| Income | |||
| Federal contribution | 365 000.00 | ||
| Contribution from Interpharma | 365 000.00 | ||
| Total contributions | 730 000.00 | ||
| Interest on bank account | 8 878.60 | ||
| Total income | 738 878.60 | ||
Expenditure | |||
| Research grants | 586 243.11 | ||
| Various 3R activities | 15 064.00 | ||
| Other sponsorship | 4 462.50 | ||
| Project supervision and information | 91 433.45 | ||
| Administrative expenses | 73 444.77 | ||
| Total expenditure | 770 647.83 | ||
| Excess expenditure over income | 31 769.23 | ||
| 770 647.83 | |||
| Balance as per 31st December 2001 | Assets | Liabilities | |
| Liquid Assets | |||
| Bank | 666 966.05 | ||
| Withholding tax | 3 107.50 | ||
| Accounting apportionment assets | 25 345.40 | ||
Liabilities | |||
| Accounting apportionment liabilities | 164 622.30 | ||
| Other liabilities | 41 000.50 | ||
| Unused project funds | |||
| Carried forward 1. 1. 2001 | 520 565.38 | ||
| expend. over income | 31 769.23 | 488 796.15 | |
| Capital of the Foundation | 1 000.00 | ||
| 695 418.95 | 695 418.95 | ||
Contingent liabilities | |||
| Approved research grants not yet paid out | 1 142 017.05. | ||
Münsingen, 17th April 2002
3R RESEARCH FOUNDATION
President: signed Dr. Hugo Wick
Secretary: signed E. Diener
As 3R Research Foundation's auditors we have examined the books and the annual financial statements (balance sheet and profit and loss account) for the year ending 31st December 2001.
The Administrative Board is responsible for drawing up the financial statements while our task is to check and assess them. We hereby confirm that we are duly qualified to do this and that we have no vested interest in the Foundation.
We have checked the accounts and statements according to the generally accepted principles of accounting, by which this task must be planned and carried out in such a way that important errors in the financial statements are reasonably certain to be identified. We checked the entries and information in the statements in an analytical and investigative way using random samples. Furthermore, we assessed the implementation of standard accounting principles, the main evaluations and the presentation of the accounts as a whole. We are of the opinion that our examination provides a solid basis for our assessment of the accounts and statements of 3R.
In our opinion, the accounts have been kept and the financial statements have been drawn up according to the relevant laws and the statutes and regulations of the Foundation.
We therefore recommend that they be approved.
Gümligen, 9th April 2002
KPMG Fides Peat
signed Markus Gasser, Cert. Auditor
signed Ursula Waber, Cert. Auditor