
3R-INFO-BULLETIN 30
| | September 2005The Authors
Prof. Dr. Hanno Würbe is head of the Animal Welfare and Ethology group at the University of Giessen (Germany). He studies environmental effects on the behaviour and well-being of animals.
The present study he conducted together with Prof. Dr. Hans-Peter Lipp, Division of Neuroanatomy and Behaviour, and Prof. Dr. Roger M. Nitsch, Division of Psychiatry Research, University of Zürich, Switzerland and their co-workers Dr. David P. Wolfer and Dr. Samuel Morf (Division of Neuroanatomy and Behaviour), Dr. Oxana Litvin (Division of Psychiatry Research). Current address: Hanno Würbel
hanno.wuerbel@vetmed.uni-giessen.de
Division of Animal Welfare and Ethology
University of Giessen
D-35392 Giessen
Germany www.uni-giessen.de/vet-tierschutz/
Editor
Peter Maier, Scientific Adviser of the 3R Research Foundation |
Environmental enrichment does not disrupt standardization
Discussions about the pros and cons of enriched housing* for laboratory rodents have a long history. Some believe that it disrupts environmental standardization, resulting in data that is less precise and less reliable, while others are convinced that it is essential for the generation of valid results. The present bulletin provides a piece of evidence to clarify the situation.
Prof. Dr. Hanno Würbel initiated a multi-laboratory study together with
two colleagues and their co-workers to compare the effects of enriched
housing in female mice on the expression of strain differences in standard
behavioural tests. The 3R Research Foundation provided substantial funding
for this study under the project no. 75-00.
Testing effects of enrichment on standardization
For many years, concerns have been raised that enriched housing[*] might disrupt standardisation by increasing variation in data obtained in animal experiments. Previous studies found variable effects of enrichment on variation in the data across variables, indicating that enrichment has no consistent effect on variation in data (see also Baumans 2003, 3R-Info-Bulletin 22). However, none of these studies provided conclusive results since they were all based on single experiments instead of several independent replicates. In contrast, we used a multi-laboratory approach involving nine independent replicates (three in each of three laboratories) to study the effects of enriched versus barren housing on (i) variation in behavioural endpoints and (ii) reproducibility of behavioural differences between three strains of mice across these independent replicates. Each replicate involved eight mice per strain and housing condition, amounting to 48 mice per replicate and 432 mice in total. Enrichment was a combination of more space, additional resources, increased environmental complexity, and novelty (novel items and environmental change). We used mice of two inbred strains (C57Bl/6J, DBA/2) and their F1-hybrids (B6D2F1). They were housed in either small barren or large enriched cages from weaning to 9 weeks of age (Fig.1a,b).

Fig. 1a: Barren housing: Makrolon type II cages with sawdust as bedding and food and water ad libitum. | 
Fig. 1b: Enriched housing: Makrolon type IV cages with sawdust, food, water and a shelter (‘Mouse House’). Twice a week one enrichment item was added, some of which were removed after one week (e.g. paper tissue, straw, shredded paper, bark, rodent pellets hidden in the sawdust), while others remained in the cage until the end of the housing period (e.g. tunnel, wooden branches, cardboard roll, cardboard house). |
Behavioral Test
At 10 weeks of age, all mice were subjected to four common behavioural tests used in drug screening studies and behavioural phenotyping of mutant mice (Fig. 2). We used identical test systems in all three labs and standardised test conditions as well as possible. Test performance in all tests was video-tracked using EthoVision 3.00 (Noldus Information Technology, Wageningen NL) and the data was analyzed using a 4-way factorial ANOVA model with housing (barren versus enriched housing), strain (DBA/2, C57Bl/6, B6D2F1), laboratory (Lipp, Nitsch, Würbel), and replicate (1, 2, 3) as between subject factors.

Fig. 2: Example of a behavioural test.
Water Maze Test: A circular pool (diameter 150 cm), filled with opaque water, containing a goal platform (14x14 cm) hidden 0.5 cm below the water surface at a constant location. The mice performed 16 training trials (4 per day) from varying start positions. On day 5, the mice performed a 60 s probe test without the goal platform.
Precision and reproducibility are maintained
To test the effects of enriched housing on the detection and reproducibility of strain differences in behaviour, we split the data by housing conditions and calculated for each replicate the proportion of variance in behavioural measures contributed by within-group variability and by laboratory x strain interactions. Figure 3 presents a synoptic summary of the results. Within-group variability contributed between 40 and 84% (average 60%) to total variance. With an average of 7.6%, the contribution of strain x laboratory interactions was considerably smaller and also less variable. However, within-group variability was unaffected by enriched housing (except for fecal bolus counts on the O-maze). This indicates that enrichment did not decrease the sensitivity of the tests to detect genetic differences and also shows that barren cages fail to reduce individual differences in behaviour. Furthermore, enrichment had no significant effect on the proportions of variance contributed by strain x laboratory interactions, and the direction of differences varied across measures, indicating that enrichment did not increase the risk of obtaining conflicting results between laboratories.
Similar to an earlier multi-lab study (Crabbe et al. 1999), we found significant strain x laboratory interactions on many variables. However, closer inspection of the data revealed that these were of quantitative rather than qualitative nature; reflecting differences in effect magnitude rather than direction of the effects (data not shown; see Wolfer et al. 2004).
The effectiveness of environmental harmonization may be limited
Between-laboratory effects (contributing on average 5.2% to total variance) and replicate effects (3.1%) made similarly small contributions to total variance, indicating that standardization between laboratories was nearly as good as standardization within laboratories. This was surprising since nothing but cage-type, enrichment protocol, light phase, test systems, and test protocols was equated across labs. It casts doubt on the effectiveness of excessive environmental harmonization to improve between-laboratory comparability of behavioural data (Würbel 2002).
More exploratory, less anxious
Enrichment had significant effects on many measures of exploration and anxiety (Fig. 3). Importantly, enriched mice showed higher exploratory activity and less anxiety-related behaviour in all three tests of exploration. Figure 4 gives an example from the Elevated O-Maze Test, indicating that enrichment effects were consistent across strains and laboratories (see Wolfer et al. 2004 for more details).
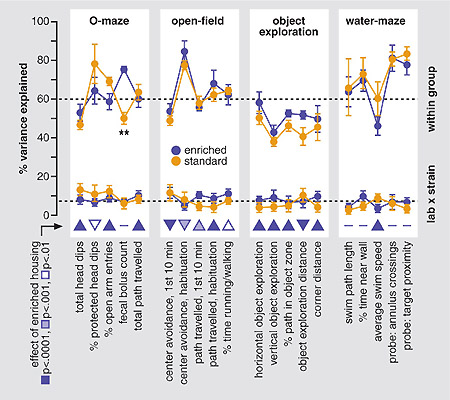
Fig. 3: Effects of enrichment on variation and reproducibility of behavioural endpoints.
Mean (± 1 s.e.) proportion of variance (%) in representative measures of the four behavioural tests contributed by within-group variability and laboratory x strain interactions. Data was pooled for the 3 strains (total N=432). (**: p<0.01). Triangles illustrate direction and significance of enrichment effects on each variable.
Beneficial for animals and research
Our findings argue against concerns that environmental enrichment might disrupt standardisation, which has hindered the implementation of enriched housing, despite its known advantages to the animals (Würbel 2001). Our findings should be generally applicable, for example to drug screening, lesion studies, and phenotyping of mutant mice. They should also apply to morphological or physiological measures, which are, in any case, less sensitive than behaviour to environmental perturbations. It remains to be seen whether our findings also apply to male mice who may sometimes respond with enhanced aggression to enrichment. At least for females, however, our results demonstrate that environmental enrichment may be used to improve animal welfare without reducing precision and reproducibility of the data, while at the same time attenuating abnormal brain function and anxiety – two potential confounds in animal experiments.
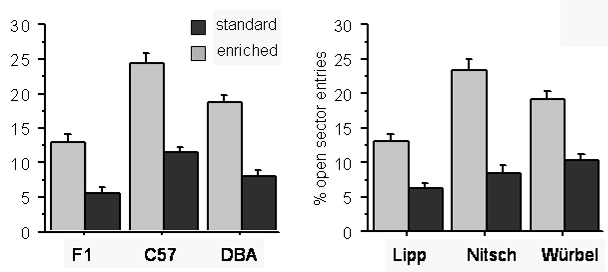
Fig. 4: Enrichment reduces anxiety in all strains and laboratories.
Effect of enrichment on mean (± 1 s.e.) proportion (%) of entries to the unprotected sectors on the Elevated O-Maze displayed by strain (left panel: data of the three labs pooled) and lab (right panel; data of the three strains pooled).
Published updated version of this Bulletin 30/2007 (PDF)
References:
- Van Praag, H., Kempermann, G. & Gage, F.H. Neural consequences of environmental enrichment. Nature Rev. Neurosci. 1, 191-198 (2000).
- Würbel, H. Ideal homes? Housing effects on rodent brain and behaviour. Trends Neurosci. 24, 207-211 (2001).
- Chapillon, P., Manneche, C., Belzung, C. & Caston, J. Rearing environmental enrichment in two inbred strains of mice: 1. Effects on emotional reactivity. Behav. Genet. 29, 41-46 (1999).
- Crabbe, J.C., Wahlsten, D. & Dudek, B.C. Genetics of mouse behavior: interactions with laboratory environment. Science 284, 1670-1672 (1999).
- Wolfer, D.P., Litvin, O., Morf, S., Nitsch, R.M., Lipp, H.P. & Würbel, H. Laboratory animal welfare: cage enrichment and mouse behaviour. Nature 432, 821-822 (2004).
- Würbel, H. Behavioral phenotyping enhanced-beyond (environmental) standardization. Genes Brain Behav. 1, 3-8 (2002).
| [*] | Barren cages: impaired brain development - abnormal behaviour - poor well-being.Environmental enrichment refers to the addition of structure and resources to barren cages with the aim to stimulate active interaction with and exploration of the environment and to facilitate the expression of species-typical behaviour. Accumulating evidence indicates that mice housed in barren laboratory cages (which are still 'standard' in many laboratories) show impaired brain development due to sensory and motor deprivation (van Praag et al. 2000); devlop abnormal repetitive behaviours (i.e. stereotypies), originating from chronically thwarted attempts to gain access to highly valued resources (e.g. shelter) or to perform highly motivated behaviours (Würbel 2001); and exhibit an anxiogenic behavioural profile compared to mice from more enriched environments (Chapillon et al. 1999). Thus, converging evidence indicates that the well-being and normal brain development of mice housed in barren cages may be seriously impaired, which also questions the validity of research done with these animals (Würbel 2001). All of these effects could be attenuated, if not eliminated, by adequate environmental enrichment, especially during early ontogeny. |






