
3R-INFO-BULLETIN 23
| | May 2003The Author
The project leader Prof. Gerd Bicker is head of a research group at the ITZ-Cell Biology, Veterinary Medical School, Hannover. His research focusses on cellular signalling by the unconventional messenger molecule nitric oxide in the nervous system of model organisms. The project was carried out together with his coworkers Francois Paquet-Durand (left) and Saime Tan (middle).
Current address: Gerd Bicker
gerd.bicker@tiho-hannover.de
Veterinary Medical
School, ITZ-Cell Biology
Bünteweg 17d
D-30559 Hannover
Germany
Editor
Peter Maier, Scientific Adviser of the 3R Research Foundation |
Simulation of stroke related damage in cultured human nerve cells.
In vivo and in vitro models are used to study cellular mechanisms during and after a stroke and to develop neuroprotective compounds that reduce brain damage after a stroke. The 3R Research Foundation supports the present successful project (Nr. 69-99) in which the differentiation procedure of a human cell line into nerve cells was optimized. This cell culture model is useful to investigate pathophysiological mechanisms involved in cerebral ischaemia. The cultured human nerve cells might replace primary cell cultures from rodent embryos and reduce the number of laboratory animals used in mechanistic experiments in stroke research and drug development.
Excitotoxic cascade during stroke
Stroke is the third leading cause of death and an important cause of adult disability in industrialised countries with their ageing human populations. Most strokes are caused by an acute interruption of the brain's blood supply, which leads to tissue ischaemia in the particularly vulnerable central nervous system. To investigate the cellular mechanisms occurring in ischaemic brain damage, a variety of rodent models[*] have been developed that mimic the pathogenic environment of nerve cells during stroke. Cell culture models are also finding increasing use (see also 3R-Info-Bulletin 15).
The brain has the highest metabolic rate of all organs and for these reasons it consumes a disproportionately high fraction of the body's oxygen and glucose. Ischaemic neurons deprived of oxygen and glucose rapidly lose their energy currency ATP, their intracellular pH then drops, and they depolarize. Among other consequences, the neurotransmitter substance glutamate is released. The accumulation of the excitatory transmitter glutamate excessively stimulates glutamate receptors of neighbouring cells, inducing increases in intracellular calcium levels and the production of free radicals, which in concert orchestrate cell injury. This cellular disorder is called excitotoxic cascade. One approach to reducing brain damage following a stroke might involve the application of neuroprotective substances that inhibit various stages of this cascade.
Generation of human nerve cells in the petri dish
Bicker's group started with a well-characterised teratocarcinoma cell line (Ntera-2, NT-2) derived from a human testicular cancer. Upon treatment with the morphogen retinoic acid, the NT-2 line can be induced to differentiate into cells that include postmitotic neurons expressing functional glutamate receptors and functional synapses, as shown by electro-physiological recordings (Ref. 1 and 2). One disadvantage of this cell culture system is the lengthy differentiation process (42 to 54 days) required until the cells reach a mature, postmitotic stage.
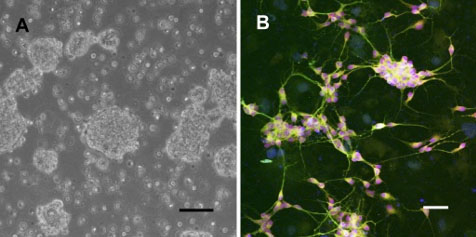
Fig. 1: Differentiation of NT-2 cells into neurons.
A Expansion of NT-2 precursors as free floating cell spheres. This technique resembles the sphere culture method used in embryonic stem cell differentiation. Scale 100 μm
B Differentiated NT-2 neurons stained by immunofluorescence. Spindle shaped cell bodies send out neural processes. Scale 50 μm
Time for differentiation is reduced by half
The conventional differentiation protocol uses adhesive substrates on which the cells are grown as monolayers. We seeded NT-2 precursor cells onto bacteriological grade petri dishes upon which these cells do not adhere. Under these conditions the cells proliferated as clusters in the shape of free floating spheres (Fig.1A). At later stages of the neuronal differentiation process, the cells were again cultured as a monolayer. Allowing the cells to proliferate as free-floating cell spheres cuts the total time needed to obtain high yields of purified NT-2 neurons to about 24 - 28 days.
The cells obtained show neuronal morphology and migrate to form ganglion-like cell conglomerates. Differentiated cells are immunoreactive to neuronal cytoskeletal markers (Fig.1B) and express the RNA-binding proteins of the ELAV-HuC/D family, which have been implicated in the differentiation of nerve cell in many organisms. Some epitheloid cells were also present in the petri dishes, but staining for neuronal markers indicated that after final plating the cell cultures were composed of approximately 80 - 95% human nerve cells (Fig.1B).
Cultured nerve cells are sensitive to ischaemic injury
In cell cultures the cell densities are lower than in intact tissue. Conversely, the volume of the surrounding medium is considerably larger than the extracellular tissue volume. Thus, only traces of oxygen are sufficient for cell survival. To mimic anoxia in culture, the NT-2 neurons were kept under an atmosphere of 95% argon and 5% CO2; to ensure complete anoxia, remaining traces of oxygen in the medium were removed with nontoxic concentrations of dithionite. Glutamate served as excitotoxin. Cell viability was assessed using the Alamar Blue assay, which measures metabolic activity. The reduction in the cells' viability over time increased with the duration of anoxia (Fig.2) and also with increasing glutamate concentration. Anoxia times of four hours resulted in the destruction of more than 80% of the cells. Most of the surviving cells did not show neuronal morphology nor did they stain positive for neuronal markers. Control experiments with undifferentiated NT-2 teratocarcinoma cells showed no vulnerability to anoxia (Ref. 3).
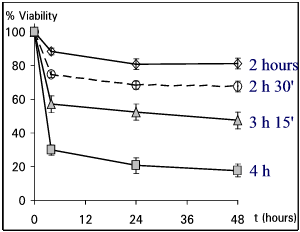 Fig. 2: Dependence of viability on duration of anoxia.
Fig. 2: Dependence of viability on duration of anoxia.
Cultured NT-2 neurons were subjected to anoxia in the presence of dithionite and 1mM glutamate. Duration of anoxia ranged from 2h to 4h. Viabilities were followed for 48 hours post anoxia treatment and plotted as percentage of internal control (100 %).
The influence of additional parameters on neuronal survival, such as type of culture medium, pH, and specific neuroactive drugs are presently being evaluated. An important parameter that influences the vulnerability of the mature postmitotic NT-2 neurons is the time in culture (Fig.3) following the 24 - 28 day differentiation process. After an additional 6 weeks in culture, NT-2 neurons are far more sensitive to ischaemic injury than "younger" neurons cultured for only 10 days, perhaps reflecting changes in the expression of neurotransmitter receptors and ion channels.
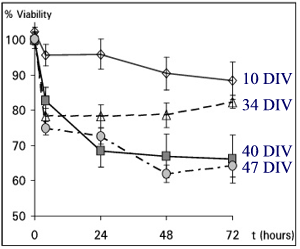 Fig. 3: Vulnerability of mature, post-mitotic NT-2 neurons depends on days in vitro (DIV) prior to anoxia treatment. Times in culture ranged from 10 to 47 days. After 2h of anoxia, percentage of viabilities was followed for 72 h.
Fig. 3: Vulnerability of mature, post-mitotic NT-2 neurons depends on days in vitro (DIV) prior to anoxia treatment. Times in culture ranged from 10 to 47 days. After 2h of anoxia, percentage of viabilities was followed for 72 h.
Reduction and replacement of laboratory animals
Stroke-related research often depends on animal models, because the interest is mainly focused on general brain damage after ischaemic lesions rather than on the effects on individual brain cells. The NT-2 cell culture system, in contrast, focuses more on the individual nerve cell or small ganglion like clusters which has a num-ber of advantages:
- NT-2 neurons are derived from a human cell line, thus they are especially suitable for screening for neuroprotective drugs effective in the human brain.
- The fairly homogeneous cellular composition of clonally derived neurons allows for large-scale cell-based assays.
- Culturing NT-2 neurons in a monolayer readily allows electro-physiological recordings and optical imaging studies to investigate the excitotoxic cascade at a cellular level. Accordingly this cell culture system might
- replace the use of primary cell cultures prepared from rodent embryos,
- reduce the number of laboratory animals needed in initial stages of drug screening and
- has the potential to replace laboratory animals in basic stroke research.
Published updated version of this Bulletin 23/2007 (PDF)
References:
- Pleasure S.J., Page C. and Lee V.M.Y. (1992) Pure, postmitotic, polarized human neurons derived from NTera 2 cells provide a system for expressing exogenous proteins in terminally differentiated neurons. J. Neurosci. 12, 1802-1815.
- Munir M., Lu L. and McGonigle P. (1995) Excitotoxic cell death and delayed rescue in human neurons derived from NT2 cells. J. Neurosci. 15, 7847-7860.
- Paquet-Durand F, Tan S. and Bicker G. (2003) Turning teratoma cells into neurons: Rapid differentiation of NT-2 cells in floating spheres. Dev. Brain Res. 142, 161-167.
| [*] | Why Animal models for ischaemic brain damage?Two rodent models are widely employed: the transient global forebrain ischaemia model, in which the entire blood supply to the brain is transiently interrupted (imitating cardiac arrest), and the focal cerebral ischaemia model, in which the proximal middle cerebral artery is occluded. In addition to the initial damage in the immediate vicinity of occluded blood vessels, cerebral ischaemia also results in a wave of delayed cell death that spreads to surrounding tissue, the so-called penumbra. Whereas neuronal death occurs rapidly in the ischaemic core, neurons remain viable for many hours in the surrounding penum-bra, providing a period of possible therapeutic intervention ("time to treatment" window). After inducing transient ischaemia, the animal is allowed to recover for some time, then is sacrificed and the brain damage studied in detail. In such studies, the bioavailability of a potential drug, its pharmacokinetics, effects on blood pressure, body temperature and motor activity are also determined. |



 Fig. 2: Dependence of viability on duration of anoxia.
Fig. 2: Dependence of viability on duration of anoxia. Fig. 3: Vulnerability of mature, post-mitotic NT-2 neurons depends on days in vitro (DIV) prior to anoxia treatment. Times in culture ranged from 10 to 47 days. After 2h of anoxia, percentage of viabilities was followed for 72 h.
Fig. 3: Vulnerability of mature, post-mitotic NT-2 neurons depends on days in vitro (DIV) prior to anoxia treatment. Times in culture ranged from 10 to 47 days. After 2h of anoxia, percentage of viabilities was followed for 72 h.