
3R-Project 121-10
Organotypic spinal cord slices to study SCI and MS like lesions
Roman Chrast1 and Josef Kapfhammer2
1Department of Medical Genetics, University of Lausanne, 1005, Lausanne, Switzerland
2Anatomical Institute, University of Basel, 4056, Basel, Switzerland
roman.chrast@unil.ch, josef.kapfhammer@unibas.ch
Keywords: mice; spinal cord; spinal cord repair / diseases; reduction; replacement
Duration: 2 years Project Completion: 2012
Background and Aim
Spinal cord injury (SCI) or diseases such as multiple sclerosis (MS) are very debilitating and need the development of new therapeutic strategies. It is a topic of intense basic and applied research. Animal models have allowed a number of findings in understanding the mechanisms involved in SCI and MS, and in testing potential therapeutic strategies. However, such in vivo models are extremely distressful for animals. Moreover, the use of a wide variety of models, and their variability and complexity, limit the ability to identify new therapies. We present a new model based on organotypic cultures of mouse spinal cord longitudinal slices (“slice model”) that we recently developed (Bonnici and Kapfhammer, 2008). This “slice model” allows to generate reproducible SCI-like and MS-like lesions and to study axonal regeneration.
Method and Results
We implemented a neuroinflammation/demyelination model on the slices by co-treating them with interferon gamma (IFNγ) and lipopolysaccharide (LPS) (Defaux et al. 2010). Using different readout including immunohistochemistry, Western blotting, qPCR, ELISA, and enzymatic assays, we characterized the features of this model and compared it to MS and SCI lesions. We observed significant demyelination with an early and progressive decrease in myelin gene expression and protein levels. Microglia was activated becoming macrophagic and proinflammatory cytokine expression and secretion were highly increased demonstrating neuroinflammation. Interestingly, astrogliosis seemed very limited. More likely as a consequence of the neuroinflammation, we also observed neurodegeneration indicated by axonal lesions, and quantitative (e.g. decrease of glutamate decarboxylase activity) and qualitative (e.g. change in phosphorylation state of neurofilament heavy chain) alterations in neuronal markers.
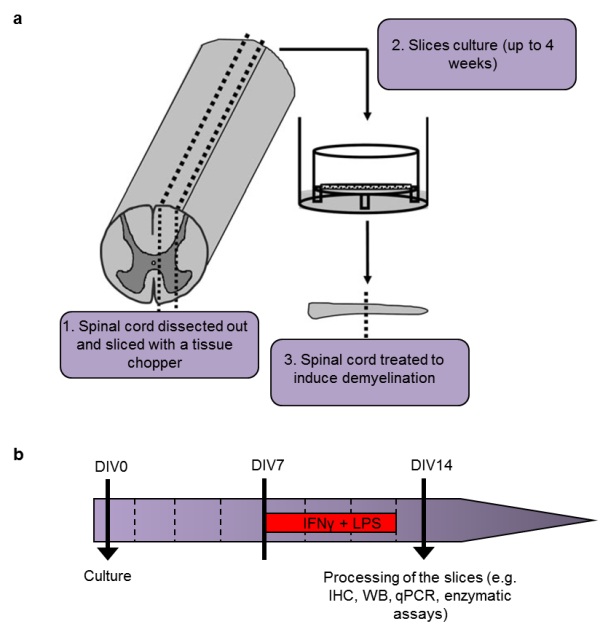
Figure 1: Induction of neuroinflammation/demyelination in our slice model.
a schematic representation of organotypic cultures of mouse spinal cord sagittal longitudinal slices (modified from Bonnici and Kapfhammer).
b) timeline summarizing the process to induce neuroinflammation/demyelination with IFNγ (100 U/ml) and LPS (10 µg/ml). Medium is changed every 2 days (dotted lines).
Conclusions and Relevance for 3R
The developed model allows in vitro reproduction of important features of both SCI and MS making it a relevant and multivalent model to study mechanisms or treatment for these diseases. Therefore, the “slice model” may substantially help to reduce animal experimentation in the field of SCI and MS where any in-vivo model is by definition extremely debilitating.
References
1. Bonnici B & Kapfhammer J, P (2008) Spontaneous regeneration of intrinsic spinal cord axons in a novel spinal cord slice culture model. European Journal of Neuroscience 27(10):2483-2492.
2. Defaux A, Zurich MG, Honegger P, & Monnet-Tschudi F (2010) Inflammatory responses in aggregating rat brain cell cultures subjected to different demyelinating conditions. (Translated from eng) Brain research 1353:213-224.
3. Lucchinetti C, et al. (2000) Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Annals of neurology 47(6):707-717.
4. Ferguson B, Matyszak MK, Esiri MM, & Perry VH (1997) Axonal damage in acute multiple sclerosis lesions. Brain 120 ( Pt 3):393-399.
5. Schirmer L, Antel JP, Bruck W, & Stadelmann C (2011) Axonal loss and neurofilament phosphorylation changes accompany lesion development and clinical progression in multiple sclerosis. Brain pathology 21(4):428-440.
6. Harrer MD, et al. (2009) Live imaging of remyelination after antibody-mediated demyelination in an ex-vivo model for immune mediated CNS damage. Experimental neurology 216(2):431-438.

