Figure 1: HCT-8 cell cultures under native conditions.

Sushila D’Souza1 and Vijay Morampudi
Pasteur Institute of Brussels-ISP, 1180-Uccle, Belgium
1present address: GSK Biologicals, Rue Fleming, 20, Wavre 1300, Belgium
Keywords: human; parasites, ectoparasites: toxoplasma gondii; epithelia; epithelia; intestine; infectious diseases; replacement; infectiosity
Duration: 2 years Project Completion: 2010
Background and Aim
The global prevalence of T. gondii, the causative agent of toxoplasmosis is estimated to be 30%. This intracellular protozoan parasite is one of the three common causes of congenital disease which can lead to severe malformation, mental retardation and spontaneous abortion. T. gondii is also the second most common cause of encephalitis in immunocompromised individuals.
Molecular epidemiological studies on a wide collection of human and animal isolates of T. gondii obtained from Europe and North America have revealed the predominance of three major clonal lineages classified as highly virulent Type I and less virulent Type II and Type III strains based on observation of mortality following inoculation of these parasites into laboratory mice.Recently, T. gondii organelles called rhoptries were shown to secrete kinases that dramatically influence host gene expression and are major parasite virulence factors (1) Our aim was to develop an in vitro cell culture based assay that would allow the early detection of T. gondii in clinical samples and would further allow an evaluation of its virulence towards the host. T. gondii infection acquired most commonly via the oral route, encounter small intestinal epithelial cells as early targats. Human small intestinal epithelial cells were therefore tested as the prototype host cell.
Method and Results
Suitable cellular hosts in vitro
An immortalized small intestinal human epithelial cell line (HCT-8) was used as in vitro target for comparative studies of the three T. gondiistrains which differ in their virulence. The cells were cultured to a monolayer (Fig. 1) and infected with an inoculum of the parasites (tachyzoites) (Figs. 2 and 3). Replication within HCT-8 cells, even at a very low multiplicity of infection, was observed of both the highly virulent Type I parasites and the low virulent Type II and Type III parasites. Type I parasites had a significantly higher rate of replication than Type II and III and this correlated with a deleterious effect on host cells, as indicated by the subsequent release of lactate dehydrogenase, a marker of host cell cytotoxicity.
Transcription factor and cytokine production
We compared the activation of NF-κB (a key transcription factor required for the activation of host genes), in HCT-8 cells by the three different T. gondiigenotype strains. Type II parasites, that displayed intermediate replication, induced higher NF-κB activity in HCT-8 cells than Type I and III parasites, confirming previous observations in human macrophages (2). We further examined if the distinct levels of NF-κB activity could be correlated to downstream cytokine production. In concordance to the high NF-κB activity observed, Type II parasites secreted significantly higher levels of IL-8 and IL-6 than Type I and Type III strains from 8h until 48h post-infection.
Expression of human β-defensin 2
Human β−defensins (HBD) are effector molecules that play an important role in early intestinal innate immune defense by their dual function primarily as antimicrobial factors to defend against pathogens and secondarily as chemotactic factors to recruit cells for adaptive immune responses (3). Thus, the differences observed in the replication capacity between the three T. gondii types could reflect differences to stimulate IEC for the secretion of antimicrobial immune effectors such as β−defensins. To investigate this possibility, we examined the early expression of antimicrobial peptide genes ΗΒD1, 2 and 3 in IEC upon infection by the three T. gondii genotype strains. A clear increase of ΗΒD2 mRNA levels occurred after 3h of infection with Type II and Type III T. gondii strains but not with Type I strains. On the contrary, no significant induction of constitutively expressed HBD1 mRNA levels was observed following infection by each of the three v genotypes. ΗΒD3 gene expression on the other hand was down-regulated by all the three T. gondii genotype strains. Our findings support the notion that virulent T. gondii parasites (Type I), unlike the less virulence strains (Type II and III), do not activate HBD2 early after infection and use this as one of the mechanism to evade early host antimicrobial effect (4).
Conclusions and Relevance for 3R
A human cell cultures based assay able to rapidly detect the presence of T. gondii in clinical samples and in addition, able to provide information on the virulence of the infecting strain would be an ideal alternative to the current test in mice and be more predictive of virulence of the parasite in humans. Comparing the pattern of cellular response of human intestinal epithelial cells, the high virulence of strain I can be distinguished from the ones with low virulence (Type II and III). The virulence is described by the combined analysis of T. gondiigrowth, cytotoxicity of target cells and the early differential response of gene expression within 3-4 hours when there is no detectable replication of the parasite and therefore no cytopathic effect (Figure 4).
(see also 3R-INFO-BULLETIN Nr. 46)
References
1. Bradley PJ and L.D. Sibley. 2007. Rhoptries: an arsenal of secreted virulence factors. Curr. Opin Microbiol. 10 (6):582-587.
2. Molestina, R. E., T. M. Payne, I. Coppens, and A. P. Sinai. 2003. Activation of NF-kappaB by Toxoplasma gondii correlates with increased expression of antiapoptotic genes and localization of phosphorylated IkappaB to the parasitophorous vacuole membrane. J Cell Sci 116:4359-4371.
3. Lai, Y., and R. L. Gallo. 2009. AMPed up immunity: how antimicrobial peptides have multiple roles in immune defense. Trends Immunol 30:131-141.
4. Morampudi, V., M. Y. Braun and S. D’Souza. 2011. Modulation of early β-defensin-2 production as a mechanism developed by Type I Toxoplasma gondii to evade human intestinal immunity. Infect Immun. 79(5):2043-2050.
Figures

Figure 1: HCT-8 cell cultures under native conditions.
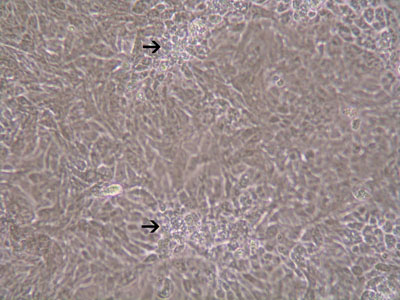
Figure 2: Plaque formation of HCT-8 cells infected with T. gondii(MOI=20) 48hrs after infection.
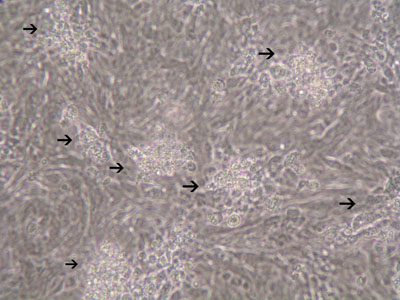
Figure 3: Increased number of plaques in HCT-8 cells infected with T. gondii(MOI=20) 72hrs after infection.
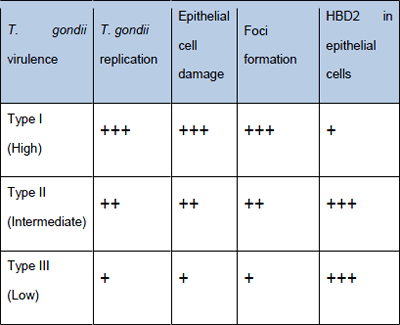
Figure 4: Cellular host (IEC) correlates of T. gondii virulence