
3R-INFO-BULLETIN 11
| | March 1999The Authors Ferber, P.C., Ferber, P.C.,
biochemist,
MD Institute of Biochemistry ETH Zurich
 Fischer, R. W., Fischer, R. W.,
biochemist Instute of Biochemistry ETH Zurich
Phone +41 1 632 31 40
Fax +41 1 632 11 21
E-mail fischer@bc.biol.ethz.ch
Editor
Greber R., Berne
|
Immunization of laboratory animals
The aim of 3R study "investigations of new adjuvants on immune response and animal distress as compared to Freund's adjuvant" was to evaluate different commercially available adjuvants in the context of hybridoma technology for the in vitro production of mouse monoclonal antibodies.
Immunization
In contrast to vaccinations in humans where antigens, mostly of viral or bacterial origin, are administered without adjuvants, the immunization of laboratory and farm animals is generally achieved by injecting the antigen together with an immune stimulating or immune potentiating adjuvant.
Freund's adjuvant
In the early fifties Freund [1] developed an adjuvant mixture that can be used universally in animals. Freund's complete adjuvant (FCA) is composed of mineral oil, heat-killed Mycobacterium tuberculosis and mannide monooleate whereas Freund's incomplete adjuvant (FIA) does not contain the Mycobacterium tuberculosis. When mixed with aqueous antigen solution, the oil in Freund's adjuvant forms a water-in-oil emulsion, and acts as an antigen depot, leading to a sustained stimulatory effect. In addition Mycobacterium tuberculosis is an immune stimulator while mannide monooleate acts as a surfactant. The currently manufactured Freund's adjuvant proved to be a potent immune stimulator but still causes severe distress to the animals. The original crude oil mix of the "classical" Freund's adjuvant, however, is no longer used since the oil manufacturing techniques of the oil producers are improved now. Nevertheless, the currently manufactured Freund's adjuvant remains a potent immune stimulator but still causes considerable distress to the animals that can be estimated by histopathological changes after injection.
Efficacy of adjuvants
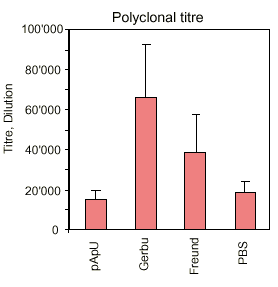
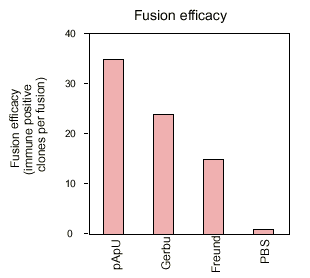
Recently immunologists have developed efficacious new adjuvants, less harmful for the animal. The efficacy of an adjuvant is normally evaluated by it's capacity to induce a polyclonal immune response in the serum of the immunized animal. This criterion of efficacy was useful for the production of polyclonal antibodies. However, the authors found it necessary to study whether in the context of hybridoma technology for in vitro monoclonal antibody production an adjuvant also produces suitable spleen cells as primary biological materials for fusion with myeloma cells. The development of new efficacious adjuvants therefore cannot uniquely be based upon polyclonal titre. The fusion efficacy of an adjuvant has to be considered as well.
Today's adjuvants
Based on a pilot study [2] in the 3R project at issue two new adjuvants were tested and compared to Freund's adjuvant: PolyApolyU (pApU, Sigma Chemical Co., St. Louis, MO), a double stranded helical ribopolynucleotide complex, known to have multiple immunological effects and used as adjuvant in human cancer therapy [3] and GERBU (GERBU Biotechnik, Gaiberg, Germany) adjuvant, a mixture of the selected components with immuno-stimulatory properties: e.g. GMDP, a synthetic analogue of MDP, the peptidoglycan responsible for the immunomodulating effect in FCA [4], Esterquats EQ1 and EQ2, Tween 80 and Cimetidine.
Experimental design
Two adjuvants (pApU and GERBU) were compared to Freund's (FCA) adjuvant. Eight different antigens (synthetic peptides, human and plant fusion proteins expressed in E.coli, and alkaline phosphatase) were used for these investigations.
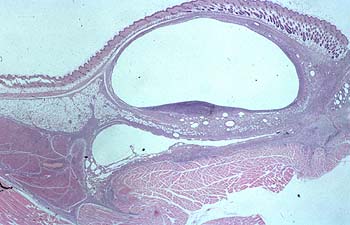
The histopathological changes at the injection sites were quantified using a four-point scale: 0 to 3 (0 = no visible changes, Fig. 1; 3 = severe changes, Fig.2).
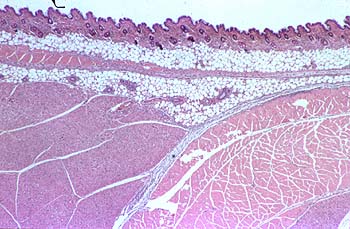 |
| Fig. 1 |
|
| Fig. 2 |
The quantification system focused more on severity than on the nature of the lesions. Extensive, diffuse infiltrations (phlegmons) were considered to be more severe than fibrotic, walled-in abscesses or granuloma. The histological analysis were carried out by P. Ossent, Institute for Veterinary Pathology University of Zurich.
|
| Fig. 3 |
|
| Fig. 4 |
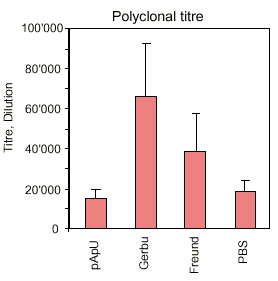
|
| Fig. 5 |
Results and conclusions
The results are summarized in a descriptive way in figures 3 - 5. It is interesting to note that the new adjuvants (pApU and GERBU) cause as little distress as the PBS which was used for control. The polyclonal titre in the serum does not necessarily correlate with the fusion efficiency which was controlled so far by bleeding before the fusion. Therefore the retro-orbital bleeding procedure can be omitted.
In conclusion, the study demonstrates that for the in vitro generation of mouse monoclonal antibodies there are available alternatives to Freund's adjuvant which are causing less distress in form of histopathological changes in the immunized animals.
Abbreviations:
EQ1: N,N-dimethyl-N,N-dihydroxyethylstearoylammonium chloride
EQ2: N-hydroxyethyl-N,N-dihydroxyethyl-stearoyl-N-methylammonium chloride
FCA: Freund's complete adjuvant
FIA: Freund's incomplete adjuvant
GMDP: N-acetyl-glycosaminyl-N-acetylmuramyl-L-alanyl-D-isoglutamine
MDP: N-acetylmuramyl-Lanalyl-D-isoglutamine
PBS: Phosphate buffered saline
References:
- Freund, J. (1951). The effect of paraffin oil and mycobacteria on antibody formation and sensitization: A review. Am. J. Clin. Pathol. 21:645-656.
- Ferber P.C., Homberger F. R., Ossent P. and Fischer R. W. (1997). Herstellung monoklonaler Antikörper: Einfluss der Adjuvantien auf die Immunantwort und die Belastung der Versuchstiere in Ersatz- und Ergänzungsmethoden zu Tierversuchen. (H. Schöffl et al. Eds) pp. 269 - 274. Springer-Verlag, Wien, New York.
- Lacour, F. (1985). Polyadenylic-polyuridylic acid: biological response-modifying activities in mice. In vivo organ distribution and pharmacokinetics in rabbits. J. Biol. Resp. Mod. 4, 490-494.
- Ellouz F., Adam A., Ciorbaru, R. and Lederer E. (1974). Minimal structural requirements for adjuvant activity of bacterial peptidoglycan derivatives. Biochem. Biophys. Res. Comm. 59, 1317-1325
 Ferber, P.C.,
Ferber, P.C., Fischer, R. W.,
Fischer, R. W.,




