
3R-Project 127-11
Establishing a novel system for quantitative production of murine basophils in vitro
Thomas Kaufmann, Institute of Pharmacology, University of Bern, 3010 Bern, Switzerland
thomas.kaufmann@pki.unibe.chKeywords: mice; transgenic mice; basophils; immunology; allergy; cell cultures: transgenic; reduction; refinement
Duration: 2 years Project Completion: 2014
Background and Aim
Basophilic granulocytes (basophils) are a rare leukocyte population, constituting ~0.5% of peripheral blood leukocytes. Recently, several high impact studies have identified previously unrecognised roles for basophils in allergic responses as well as in immune regulation, indicating that the role of basophils may have been underestimated so far (1-5) The study of basophils is particularly challenging as no suitable cell culture model for human or mouse basophils exists to date. Moreover, in vitro differentiation of basophils from bone marrow is very inefficient. Therefore, large numbers of mice have to be sacrificed in order to isolate very limited numbers of primary basophils from blood.
For those reasons, we have recently established a novel method to generate conditionally immortalised, basophil-committed progenitor cell lines, which can be differentiated in vitro in near-unlimited amounts into mature basophils. The main aim of the 3R project is the full phenotypical and functional characterisation of this, to our knowledge, unique cellular mode for murine basophils.
Method and Results
Basophil-committed progenitor cell lines are generated from murine bone marrow (or foetal livers) using conditional Hoxb8 by a significantly modified protocol published for the quantitative production of mouse neutrophils/macrophages (6). The most important changes include the use of different cytokines and a novel lentiviral system for conditional Hoxb8 expression. (7) Once immortal cells lines are established, they can be easily handled and manipulated and maintained in culture for prolonged times (Figure 1A). Differentiation into mature basophils is achieved 'on demand' by shutdown of exogenous Hoxb8 expression (Figure 1B). Within 6 days, an enriched population of cells resembling mature basophils is obtained. Mature Hoxb8 basophils are end-differentiated (non-cycling), have lobulated nuclei and a granular appearance and display a surface expression profile of a number of membrane bound, cell type specific surface proteins (FcεRIhighCD11b+ckitnegGr1negIL5RnegCCR3neg, see Figure 1C, D and not shown). The high affinity receptor for IgE, FcεRI, is functional, as receptor crosslinking leads to degranulation and release of N-acetyl-β-D-hexosaminidase (Figure 1E).
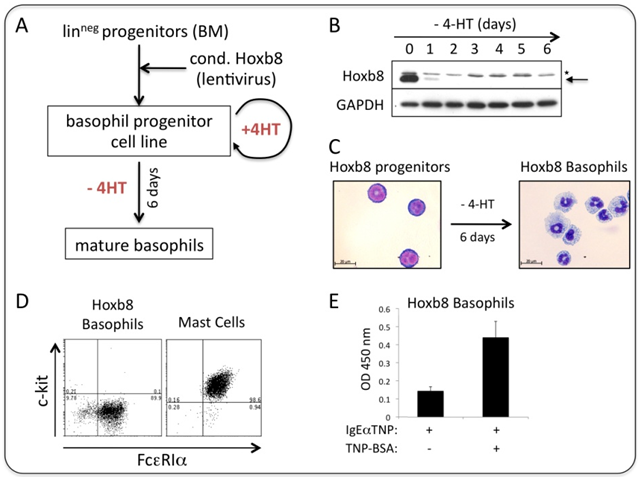
Figure 1: Generation of murine basophils in vitro using conditional Hoxb8
(A) Simplified scheme of generation of basophil committed progenitor cell lines from bone marrow.
(B) Western blot showing rapid shutdown of exogenous Hoxb8 upon removal of 4-hydroxytamoxifen (4HT).
(C) DiffQuik staining of immature and mature Hoxb8 basophils.
(D) c-kit (CD117) and FcεRI surface expression profile by FACS of mature Hoxb8 basophils and bone marrow derived mast cells.
(E) N-acetyl-ε-D-hexosaminidase release (as a measure for degranulation) upon FcεRI crosslinking.
Phenotypical and functional characterisation was performed in immature progenitors as well as differentiated basophils. We investigated the expansion and clonogenic potential of Hoxb8 basophils and extended the characterisation of basophil specific surface markers and enzymes (e.g. mast cell protease 8 and 11, see Ugajin et al.8). Functionality of Hoxb8 basophils was tested by crosslinking FceRI9,10 followed by the analysis of induction and/or release of lipid mediators (e.g. leukotriene C4), histamine and cytokines (IL-4, IL-13). Treatment with the protein fragment C5a (released from complement component C5) was used in parallel as an immunoglobulin-independent stimulus. Furthermore, chemotaxis, transmigration behaviour and oxidative burst upon activation with various stimulants was or is being investigated.
Conclusions and Relevance for 3R
We have established a method to generate basophil-committed progenitor cell lines using conditional Hoxb8. These cells can be differentiated in quantitative amounts into mature basophils in vitro, making it, to our knowledge, a novel and unique cellular model for murine basophils. The basophil lines described here are genetically stable, can be generated from any genetically modified mouse strain and can easily be manipulated (e.g. knockdown by short-hairpin RNA, introduction of transgene, etc.) and progenitors can be differentiated into mature basophils in near unlimited numbers. As large amounts of mice are required to isolate primary basophils, we would like to promote this cellular model as an alternative method to study basophil biology, in particular to answer questions requiring large numbers of basophils (e.g. biochemical work, study of signalling pathways, etc.). Additionally, Hoxb8 basophil lines can be generated form foetal livers (E12-E14) of mouse strains displaying severe phenotypes. The main relevance for 3R of this project therefore lies in its potential to reduce the number of sacrificed mice and the need to grow mice with severe phenotypes to adulthood.
Publications
i) directly related
- Gurzeler U, Rabachini T, Salmanidis M, Brumatti G, Ekert PG, Echeverry N, Bachmann D, Simon HU, and Kaufmann T. In vitro differentiation of near-unlimited numbers of functional mouse basophils using conditional Hoxb8, Allergy (2013) 68: 604–613; highlighted in editorial (Gibbs & Nilsson, same issue).
- Reinhart R, Wicki S, and Kaufmann T. In vitro differentiation of mouse granulocytes, Methods Mol Biol (2016),1419:95-107.
ii) subsequent publications using “Hoxb8 basophils”
- Morshed M, Hlushchuk R, Simon D, Walls AF, Obat-Ninomiya K, Karasuyama H, Djonov V, Eggel A, Kaufmann T, Simon HU, and Yousefi S. NADPH oxidase-independent formation of extracellular DNA traps by basophils, J Immunol (2014), 192: 5314-5323.
- Yousefi S, Morshed M, Amini P, Stojkov D, Simon D, von Gunten S, Kaufmann T, and Simon HU. Basophils exhibit antibacterial activity through extracellular trap formation, Allergy (2015) 70(9):1184-8.
- Reinhart R, Rohner L, Wicki S, Fux M, and Kaufmann T. BH3 mimetics efficiently induce apoptosis inn mouse basophils and mast cells.Cell Death Diff, doi:10.1038/cdd.2017.154. [Epub ahead of print].
References
1. Karasuyama H, Mukai K, Tsujimura Y, Obata K. Newly discovered roles for basophils: a neglected minority gains new respect. Nat Rev Immunol 2009 Jan; 9 (1): 9-13.
2. Mukai K, Matsuoka K, Taya C, Suzuki H, Yokozeki H, Nishioka K, et al. Basophils play a critical role in the development of IgE-mediated chronic allergic inflammation independently of T cells and mast cells. Immunity 2005 Aug; 23 (2): 191-202.
3. Obata K, Mukai K, Tsujimura Y, Ishiwata K, Kawano Y, Minegishi Y, et al. Basophils are essential initiators of a novel type of chronic allergic inflammation. Blood 2007 Aug 1; 110 (3): 913-920.
4. Oh K, Shen T, Le Gros G, Min B. Induction of Th2 type immunity in a mouse system reveals a novel immunoregulatory role of basophils. Blood 2007 Apr 1; 109 (7): 2921-2927.
5. Tsujimura Y, Obata K, Mukai K, Shindou H, Yoshida M, Nishikado H, et al. Basophils play a pivotal role in immunoglobulin-G-mediated but not immunoglobulin-E-mediated systemic anaphylaxis. Immunity 2008 Apr; 28 (4): 581-589.
6. Wang GG, Calvo KR, Pasillas MP, Sykes DB, Hacker H, Kamps MP. Quantitative production of macrophages or neutrophils ex vivo using conditional Hoxb8. Nat Methods 2006 Apr; 3 (4): 287-293.
7. Vince JE, Wong WW, Khan N, Feltham R, Chau D, Ahmed AU, et al. IAP antagonists target cIAP1 to induce TNFalpha-dependent apoptosis. Cell 2007 Nov 16; 131 (4): 682-693.
8. Ugajin T, Kojima T, Mukai K, Obata K, Kawano Y, Minegishi Y, et al. Basophils preferentially express mouse Mast Cell Protease 11 among the mast cell tryptase family in contrast to mast cells. J Leukoc Biol 2009 Dec; 86 (6): 1417-1425.
9. Ekoff M, Kaufmann T, Engstrom M, Motoyama N, Villunger A, Jonsson JI, et al. The BH3-only protein Puma plays an essential role in cytokine deprivation induced apoptosis of mast cells. Blood 2007 Nov 1; 110 (9): 3209-3217.
10. Marsden VS, Kaufmann T, O'Reilly L A, Adams JM, Strasser A. Apaf-1 and Caspase-9 are required for cytokine withdrawal-induced apoptosis of mast cells but dispensable for their functional and clonogenic death. Blood 2006 Nov 15; 107 (5): 1872-1877.

