
3R-Project 106-07
Standardization and Pre-validation of MucilAir: A novel in vitro cell model of the human airway epithelium for testing acute and chronic effects of chemical compounds
Song Huang
Epithelix Sàrl, 1228 Plan-les-Ouates, Geneva, Switzerland
song.huang@epithelix.com
Keywords: human; epithelia; epithelia; lung; toxicology; cell cultures: 3d cultures; replacement; toxicity testing: aerosols; validation
Duration: 1 year Project Completion: 2009
Background and Aim
Even though the alternative methods and models for addressing respiratory problems exist (Farmen et al., 2005; Matsui et al., 2005; Ostedgaard et al., 2005; Tarran et al., 2006; Geiser-Kamber, 2004; Kalberer et al., 2006), none of them have gone through the validation process. In this situation, it would be very difficult for industrial partners to use these alternative methods: Due to the legal constraints, the chemical industry, for example, cannot register products which are not tested with validated methods. As a result, animals will still be used in the future. With the implementation of REACH, more than 30’000 chemicals will have to be tested within the next 11 years. Thus, the standardization and validation of alternative methods is becoming a matter of urgency.
The compagny Epithelix, has developed and marketed a novel in vitro cell model of the human airway epithelium (MucilAir) which has some unique features:
- It is of human origin and primary cell culture
- It closely mimics the morphology and functions of the normal human airway epithelium
- It has a unique shelf-life of one year
- It is easy to handle and maintain
- Adapted to high-throughput screening and testing
- Serum free
These features of our model make it now possible to perform short-term and long-term toxicity tests using an in vitro cell model. The aim of this project is to collect enough experimental data on MucilAir so a dossier for pre-validation could be submitted to ECVAM.
Method and Results
According the ECVAM principles on test validity (Curren et al., 1995; Hartung et al., 2004), in order to validate an alternative method, a pre-validation step must be performed. This step includes several criteria:
1: Test definition:
- A definition of the scientific purpose of the test:
Test of the acute and chronic toxicity of the chemical compounds by inhalation.
- A description of the mechanistic basis of the test in view of the broader current scientific knowledge of the test endpoint: MTT, Resazurin test and TEER
- A definition of the protocol compliant with the Good Laboratory Practice.
2: Within-laboratory variability:
this addresses the variability over time and for different operators, but using the same laboratory set-up. Concretely, we propose to work out the following end-points:
a) Basal cytotoxicity tests: MTT and Resazurin test.
There are several traditional endpoints for evaluating the cytotoxic effects of chemical compounds, such as the MTT, Resazurin test. The protocols for these two tests are well established.
b) Integrity of the epithelium: Trans-epithelial Electric Resistance (TEER): Cellular electrophysiological properties, such as trans-epithelial resistance, are excellent indicators of the integrity of the epithelium. Furthermore, the TEER measurement is a non-destructive method. Therefore, it can be used as an endpoint alone for long-term toxicity tests or combined with other endpoints (MTT, Resazurin).
The preliminary results of cytotoxicity tests:
A: Reproducibility:
In order to perform routine toxicity tests, measurable end-points have to be defined. We chose the MTT measurement as an endpoint evaluating the cyto-toxicity effects of the chemical compounds. Preliminary tests have been performed on MucilAir to establish a standard protocol, as well as to assess reproducibility. A protocol has been established. The results are reproducible when tested on 24 inserts, with a very small standard deviation (see Table in Figure 1).
B: Relevance - Cytotoxic Effect of Acrolein on MucilAir:
Next, we tested the cyto-toxic effects of Acrolein on MucilAir. The results obtained (Fig. 2) correlated very well with the published data (Haddad-Romet, 1993, Official Data of OCDE).
Conclusions and Relevance for 3R
Actually, there is no alternative method for testing the chronic toxicity chemicals. All tests have to be done on animals such as rats. We would like to offer a validated alternative method/model which will have profound implications in relation to 3R’s animal protection principles.
First, unlike most of the airway epithelial cell models which can survive for only several days or weeks, our in vitro cell model has a shelf-life of one year. This unique feature makes it possible now to test the chronic effects of drugs or chemicals.
Secondly, the mission of Epithelix is not research per se; rather, Epithelix is actively applying and promoting the 3R principles by offering a ready-for-use in vitro cell model as a full package. Therefore, the impact on the animal experimentations is not limited to one research laboratory as it was often the case; instead, its impact is worldwide. If its full potential could be reached, then the life of a lot of animals might be saved.
(see also 3R-INFO-BULLETIN Nr. 41)
References
Curren RD, Southee JA, Spielmann H, Liebsch M, Fentem JH and Balls M. The role of prevalidation in the development, validation and acceptance of alternative methods. ATLA 23, 211-217, March/April 1995
Geiser-Kamber M: 3R-Project 89-03In vitro replica of the inner surface of the lungs, for the study of particle-cell interaction.
Haddad-Romet S
L’Épithélium Respiratoire en culture pour l’évaluation toxicologique
OPAL, Edition de Septembre 1993, ISBN: 2-87914-015-3. ISSN: 1250-5536
Hartung T, Bremer S, Casati S, Coecke S, Corvi R, Fortaner S, Gribaldo L, Halder M, Hoffmann S, Roi AJ, Prieto P, Sabbioni E, Scott L, Worth A, Zuang V. A modular approach to the ECVAM principles on test validity. Altern Lab Anim. 2004 Nov; 32(5):467-72.
Kalberer M., Savi M., Lang D., Burtscher H., Fierz M., Mohr M. and Geiser M. (2006) On-line deposition chamber for organic aerosols onto lung epithelial cell cultures - deposition characteristics and first results of morphological analysis and cellular responses. (Abstract, Int. Aerosol Conference, Minnesota 2006).
Matsui H, Verghese MW, Kesimer M, Schwab UE, Randell SH, Sheehan JK, Grubb BR, Boucher RC. Reduced three-dimensional motility in dehydrated airway mucus prevents neutrophil capture and killing bacteria on airway epithelial surfaces. J. Immunol. (2005) Jul 15; 175(2):1090-9.
Ostedgaard LS, Rokhlina T, Karp PH, Lashmit P, Afione S, Schmidt M, Zabner J, Stinski MF, Chiorini JA, Welsh MJ. A shortened adeno-associated virus expression cassette for CFTR gene transfer to cystic fibrosis airway epithelia. Proc Natl Acad Sci U S A. 2005 Feb 22; 102(8):2952-7
Tarran R, Button B, Boucher RC. Regulation of normal and cystic fibrosis airway surface liquid volume by phasic shear stress. Annu Rev Physiol.; 2006, 68:543-61.
Vermeer PD, Einwalter LA, Moninger TO, Rokhlina T, Kern JA, Zabner J and Welsh M.
Segregation of receptor and ligand regulates activation of epithelial growth factor receptor.
Nature 20 MARCH 2003, VOL 422, 322-326.
Wiszniewski L, Jornot L, Dudez T, Pagano A, Rochat T, Lacroix JS, Suter S, Chanson M. : Long-term cultures of polarized airway epithelial cells from patients with cystic fibrosis.
Am J Respir Cell Mol Biol. 2006 Jan; 34(1):39-48. Epub 2005 Sep 22.
Wiszniewski L, Sanz J, Scerri I, Gasparotto E, Dudez T, Lacroix JS, Suter S, Gallati S, Chanson M: Functional expression of connexin30 and connexin31 in the polarized human airway epithelium. Differentiation, 2007, in press.
Figures

Figure 1: To test the reproducibility of the MTT test on MucilAir, 24 inserts of fully differentiated airway epithelium were submitted to MTT test, using a protocol that we optimized for MucilAir. As shown above, the MTT test using MucilAir is highly reproducible and reliable.
Figure 2
Cytotoxicity effects of Acrolein, a well-known toxic compound for the airway epithelium, were tested on MucilAir (incubation period of 24h). The “dose-effect” response is between 0.5 and 3.0 mM. The EC50 obtained (1.5 mM) was in good correlation with the published results (N=24).
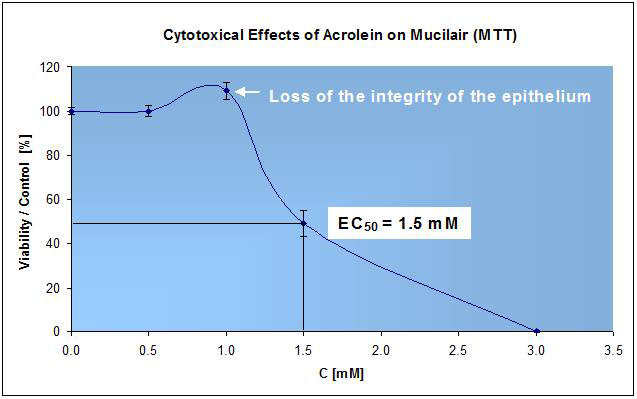
Figure 2


