 |
de | fr | en Druckansicht ![]()
3R-Project 79-01
Screening methods for repellents and attachment deterrents for ticks in-vitro
Patrick Guerin and Thomas Kröber
Institute of Biology, University of Neuchâtel, 2007 Neuchâtel, Switzerland
patrick.guerin@unine.ch, thomas.kroeber@unine.ch
Keywords: tick; veterinary drugs; veterinary drugs; reduction; replacement; diagnostic approaches: parasites; veterinary drugs
Duration: 2 years Project Completion: 2003
Background and Aim
Because many insects have developed a resistance to pesticides, new strategies, such as the development of repellents and attachment deterrents, are required to protect people and animals from blood-sucking ectoparasites. The prevention of tick bites is a particularly important focus of such research. Ticks transmit Lyme disease and a range of other pathogens to humans and animals during their blood meal. The economic impact of ticks in livestock farming (free-ranging cattle and other domestic animals) is significant. Another current key area of work is the control of ticks on pets.
Repellents and attachment deterrents are designed to disrupt the normal host-parasite relationship and inhibit the parasite's ability to suck blood from its host. Protection results in reduced loss of blood by hosts and reduced incidence of diseases transmitted. Our aim is to develop realistic in-vitro methods to screen repellents and attachment deterrents for ticks in order to reduce the need for mass screenings in live animals. We wish to bring our work on in-vitro assays to the attention of relevant practitioners at the start of the project in order to establish working contacts.
Method and Results
This project is based on our knowledge of the chemosensory pathways leading to host selection, attachment, feeding and mating in a range of tick species. Natural products that influence these behaviour patterns have been identified by in-vitro assays (http://www.unine.ch/zool/commun/articles.htm). The novelty of our approach is to measure the extent to which candidate repellents and attachment deterrents can inhibit in-vitro the effects of chemical stimuli inadvertently offered by the host. Only those products that can overcome the effects of the host's stimuli can be considered as effective repellents and attachment deterrents for protection of humans and animals.
We have three in-vitro test methods (Fig.1-3): i) a locomotion compensator that records the tick's movement towards test attractants from animals and the effects of repellents on this movement, ii) a test with an "artificial arm", and iii) artificial feeding membranes that simulate the skin, allowing attachment deterrents to be examined (see also 3R Project 10-88).
The final phase of the project is reserved for communicating the findings at scientific and industrial meetings, and to transfer the in-vitro methods to the relevant end users.
Conclusions and Relevance for 3R
The proposed work will demonstrate that mass screenings for potential repellents and attachment deterrents for ticks can be carried out reliablyin-vitro. By demonstrating the potential of the in-vitro assays for mass screening it is our aim that end users will be stimulated to switch from animal models to cheaper and more standardised in-vitro alternatives. To achieve the reduce/replace goal, an essential ingredient will be to work with practitioners from the outset by inviting them to submit potentially useful products of their own to demonstrate their efficacy in the in-vitro assays we are developing.
(see also 3R-INFO-BULLETIN Nr. 27)
Published updated Version 27/2007 (pdf)
References
1) Guerin PM, Kröber T, McMahon C, Guerenstein P, Grenacher S, Vlimant M, Diehl PA, Steullet P and Syed Z, (2000) Chemosensory and behavioural adaptations of ectoparasitic arthropods. Novo Acta Leopoldina 83 213-229.
2) McMahon C., Kröber T. and Guerin P.M. (2003) In vitro assays for repellents and deterrents for ticks: differing effects of products when tested with attractant ar arrestment stimuli. Medical and Veterinary Entomology 17:370-378.
3) Kröber T. and Guerin P.M. (2007) An in vitro feeding assay to test acaricides for control of hard ticks. Pest Management Sciences 63: 17-22.
4) Kröber T. and Guerin P.M. (2007) In vitro feeding assays for hard ticks. TRENDS in Parasitology 23 / 9: 445-449.
see ScienceDirect, open access articlesee
Figures
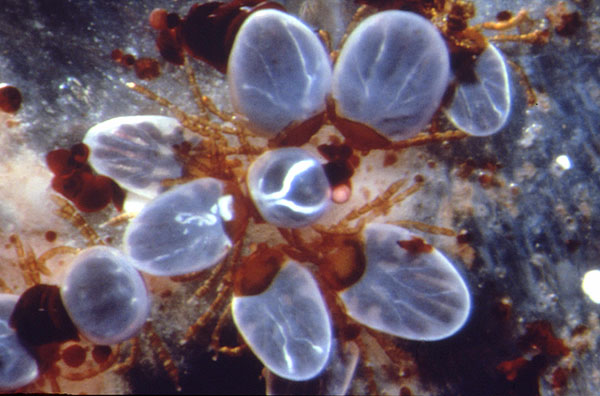
Figure 1: Mature larvae of the cattle tick Boophilus microplus feeding on blood through an artificial membrane. Most larvae are replete with blood. Note the brown faeces excreted by the ticks on the membrane. (Photo F. Kuhnert)
Figure 2
The larval stage of the cattle tick Boophilus microplus (total size 0.3 mm) has sensillae with chemoreceptors on the tips of its legs and mouth parts (blue arrows). The tick uses its sensillae to regularly probe the surfaces it walks on and in this way obtain information about the chemical nature of the substrate.
Figure 3
Larvae from the cattle tick Boophilus microplus aggregating in the quarter (upper left) of a Petri dish floor treated with steer hair extract.

Figure 2
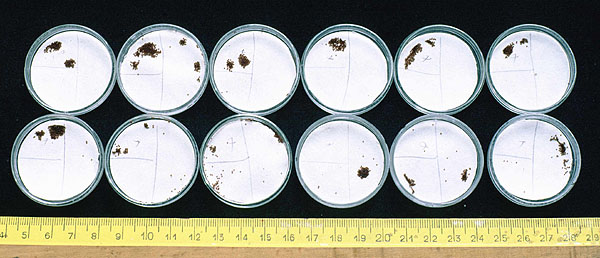
Figure 3
| Letzte Änderung: 12.10.2018 |